
Concept explainers
(a)
Interpretation:
The compound
Concept Introduction:
Naming molecular compounds:
To name binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
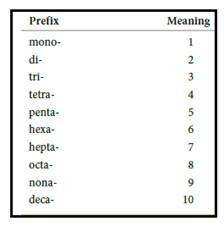
Figure 1
(b)
Interpretation:
The compound
Concept Introduction:
Binary oxygen containing compound are categorised on the basis of oxidation state of their oxide in the compound. A binary compounds has Oxygen with oxidation number of
(c)
Interpretation:
The compound
Concept Introduction:
Naming molecular compounds:
To name binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
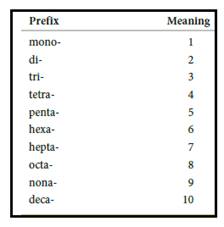
Figure 1
(d)
Interpretation:
The compound
Concept Introduction:
Binary oxygen containing compound are categorised on the basis of oxidation state of their oxide in the compound. A binary compounds has Oxygen with oxidation number of
(e)
Interpretation:
The compound
Concept Introduction:
Binary oxygen containing compound are categorised on the basis of oxidation state of their oxide in the compound. A binary compounds has Oxygen with oxidation number of
(f)
Interpretation:
The compound
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
General Chemistry: Atoms First
- What is the relationship between the following two compounds?arrow_forwardThe analysis of the structure of compound X which has the molecular formula C14H10O2 provides spectroscopic data as shown below. By systematic analysis, determine the structure of the compound!arrow_forwardA medical examiner is trying to determine the cause of death of a 48 year old male, Archie, who seems to have been poisoned. Police found rat poison (Strychnine - C21H22N2O2) in the home, so one suspect is his wife Lisa. However, upon questioning Lisa, she blames their neighbor Ralph, an anthropologist who studies antique South American hunting arrows. These arrows happen to contain the deadly poison, Curare (C40H44N4O), so Ralph is also being considered a suspect. An analysis of Archie's blood was done using mass spectrometry and a compound that contains 80.66% carbon, 7.39% hydrogen, 9.39% nitrogen, and 2.68% oxygen was found. Can you determine the empirical formula for the compund to determine if it could match one of these two poisons?arrow_forward
- Name the following compounds and answer the sub-questions that follow:arrow_forward6. Give the structural formulas of three compounds of the composition С5Н10О2 belonging to different classes of organic compounds.arrow_forwardName the following compounds:(a) NaF(b) Rb2O(c) BCl3(d) H2Se(e) P4O6(f) ICl3arrow_forward
- what is the relationship between the following compounds?arrow_forwardcan someone help me please,and explain what is going on, I'm confused for the first recaction I was going to use PBr3 and then for the second part add C6H12O.arrow_forwardFor the following reactions of one mole ofreactant, predict the product, including its structuralformula and name, and state the chemical amount (inmoles) of hydrogen required for complete reaction. HCCCH2CH2C6H5 + ___ H2(g) →arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
