
Package: Organic Chemistry with Connect 2-year Access Card
5th Edition
ISBN: 9781259729959
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 19.17P
Rank the compounds in each group in order of increasing acidity.
a.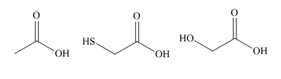
b.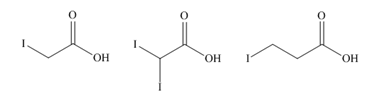
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the compounds in each group in order of increasing acidity.
The order of increasing acidity is
Phenol < meta-cyanophenol < ortho-cyanophenol
1.how about para-cyanophenol??does it has higher acidicity than both ortho-cyanophenol and meta cyanophenol or lower??
2.why does ortho-cyanophenol have higher acidicity than meta cyanophenol??
3. why doesmeta cyanophenol have higher acidicity than Phenol
Rank the following compound in each group based on increasing acidity
Chapter 19 Solutions
Package: Organic Chemistry with Connect 2-year Access Card
Ch. 19 - Prob. 19.1PCh. 19 - Problem 19.2 Give the structure corresponding to...Ch. 19 - Problem 19.3 Draw the structure corresponding to...Ch. 19 - Prob. 19.4PCh. 19 - Prob. 19.5PCh. 19 - Problem 19.6 Rank the following compounds in order...Ch. 19 - Problem 19.7 Explain how you could use IR...Ch. 19 - Prob. 19.8PCh. 19 - Problem 19.9 How many tetrahedral stereogenic...Ch. 19 - Problem 19.10 What alcohol can be oxidized to each...
Ch. 19 - Problem 19.11 Identify A-D in the following...Ch. 19 - Problem 9.12 Draw the cation that results when a...Ch. 19 - Problem 19.13 Draw the products of each acid-base...Ch. 19 - Problem 19.14 Given the values in Appendix A,...Ch. 19 - Problem 19.15 Rank the labeled protons in...Ch. 19 - Problem 19.16 Match each of the following values ...Ch. 19 - Problem 19.17 Rank the compounds in each group in...Ch. 19 - Problem 19.18 Rank the compounds in each group in...Ch. 19 - Prob. 19.19PCh. 19 - Problem 19.20 Which of the following pairs of...Ch. 19 - Problem 19.21 Two other commonly used sulfonic...Ch. 19 - Problem 19.22 Draw both enantiomers of each amino...Ch. 19 - Problem 19.23 Explain why amino acids, unlike most...Ch. 19 - Problem 19.24 Draw the positively charged,...Ch. 19 - Prob. 19.25PCh. 19 - Problem 19.26 Explain why the of the group of...Ch. 19 - Answer each question for A and B depicted in the...Ch. 19 - Prob. 19.28PCh. 19 - 19.29 Give the IUPAC name for each compound.
a....Ch. 19 - 19.30 Draw the structure corresponding to each...Ch. 19 - Prob. 19.31PCh. 19 - 19.32 Rank the following compounds in order of...Ch. 19 - 19.33 Draw the organic products formed in each...Ch. 19 - 19.34 Identify the lettered compounds in each...Ch. 19 - 19.35 Using the table in Appendix A, determine...Ch. 19 - 19.36 Draw the products of each acid-base...Ch. 19 - 19.37 Which compound in each pair has the lower ?...Ch. 19 - 19.38 Rank the compounds in each group in order of...Ch. 19 - 19.39 Rank the compounds in each group in order of...Ch. 19 - 19.40 Match the values to the appropriate...Ch. 19 - Prob. 19.41PCh. 19 - 19.42 Which carboxylic acid has the lower ,...Ch. 19 - Prob. 19.43PCh. 19 - 19.44 Explain the following statement. Although...Ch. 19 - Prob. 19.45PCh. 19 - 19.46 Explain why the of compound A is lower than...Ch. 19 - 19.47 Rank the following compounds in order of...Ch. 19 - Prob. 19.48PCh. 19 - Prob. 19.49PCh. 19 - 19.50 Draw all resonance structures of the...Ch. 19 - As we will see in Chapter 23, CH bonds are...Ch. 19 - Prob. 19.52PCh. 19 - The pKa of acetamide (CH3CONH2) is 16. Draw the...Ch. 19 - 19.54 Write out the steps needed to separate...Ch. 19 - Prob. 19.55PCh. 19 - Can octane and octan -1- ol be separated using an...Ch. 19 - 19.57 Identify each compound from its spectral...Ch. 19 - 19.58 Use the NMR and IR spectra given below to...Ch. 19 - 19.59 An unknown compound (molecular formula )...Ch. 19 - 19.60 Propose a structure for (molecular formula...Ch. 19 - 19.61 Match the data to the appropriate...Ch. 19 - Prob. 19.62PCh. 19 - Prob. 19.63PCh. 19 - Prob. 19.64PCh. 19 - 19.65 For each amino acid ,draw its neutral,...Ch. 19 - Calculate the isoelectric point for each amino...Ch. 19 - 19.67 Lysine and tryptophan are two amino acids...Ch. 19 - Prob. 19.68PCh. 19 - Prob. 19.69PCh. 19 - Prob. 19.70PCh. 19 - 19.71 Hydroxy butanedioic acid occurs naturally in...Ch. 19 - 19.72 Although it was initially sold as a rat...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Rank the compounds in each group in order of increasing basicity.arrow_forwardCaftaric acid is found in grapes, wine, and raisins. Rank the labeledprotons in caftaric acid in order of increasing acidity.arrow_forwardRank the following compounds in order of increasing acidity, and explain in detail your choice of order.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY