
(a)
Interpretation:
The curved arrow mechanism for the given reaction is to be stated.
Concept introduction:
The curved-arrow notation is used to show the transfer of electrons from one atom to another. The curved arrow has two barbs (head and tail) which represent the direction of electron flow.
Answer to Problem 19.64AP
The curved arrow mechanism for the given reaction is shown below.
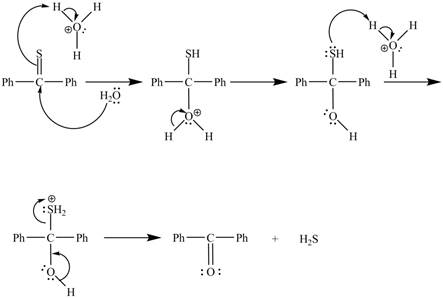
Explanation of Solution
The given reaction is shown below.
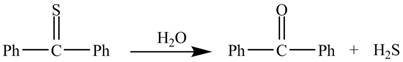
Figure 1
In the above reaction, diphenylmethanethione undergoes hydrolysis to yield benzophenone.
The curved arrow mechanism for the reaction is shown below.
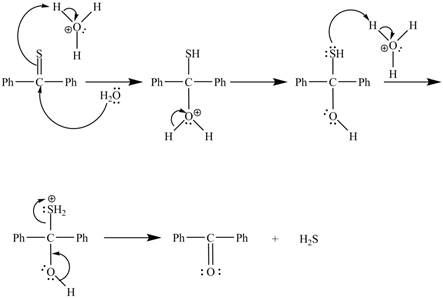
Figure 2
In the above mechanism, lone pairs of water attacks the
The curved arrow mechanism for the reaction is shown in Figure 2.
(b)
Interpretation:
The curved arrow mechanism for the given reaction is to be stated.
Concept introduction:
The curved-arrow notation is used to show the transfer of electrons from one atom to another. The curved arrow has two barbs (head and tail) which represent the direction of electron flow.
Answer to Problem 19.64AP
The curved arrow mechanism for the given reaction is shown below.
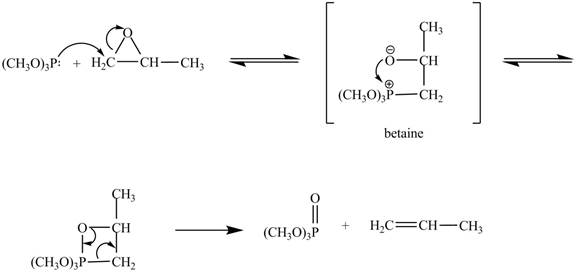
Explanation of Solution
The given reaction is shown below.

Figure 3
In the above reaction, trimethylphosphite reacts with
The curved arrow mechanism for the given reaction is shown below.
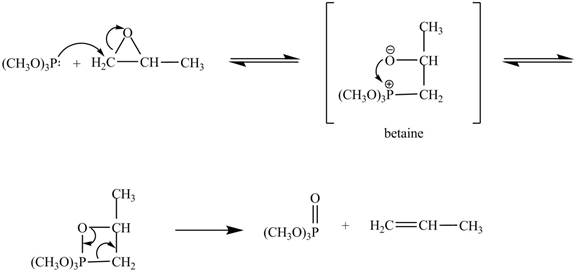
Figure 4
In the above mechanism, the trimethylphosphite reacts the least hindered site of the
The curved arrow mechanism for the given reaction is shown in Figure 4.
(c)
Interpretation:
The curved arrow mechanism for the given reaction is to be stated.
Concept introduction:
The curved-arrow notation is used to show the transfer of electrons from one atom to another. The curved arrow has two barbs (head and tail) which represent the direction of electron flow.
Answer to Problem 19.64AP
The curved arrow mechanism for the given reaction is shown below.
Explanation of Solution
The given reaction is shown below.
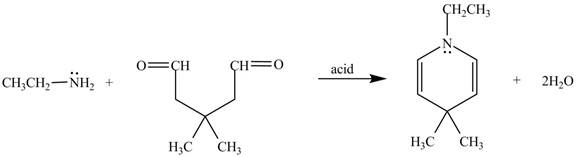
Figure 5
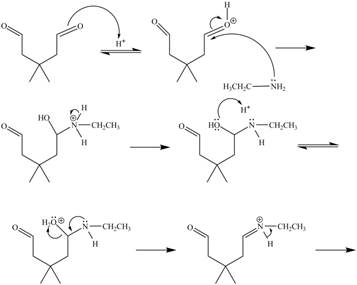
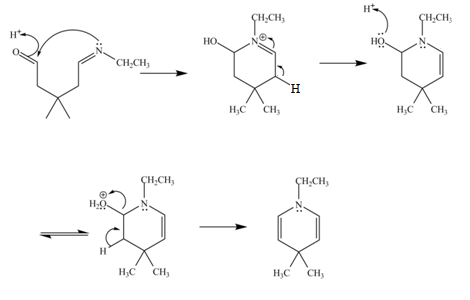
In the above reaction, ethylamine reacts with
The curved arrow mechanism for the given reaction is shown below.
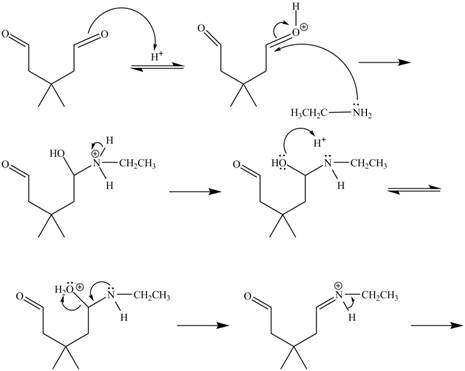

Figure 6
In the above mechanism, the carbonyl carbon of
The curved arrow mechanism for the given reaction is shown in Figure 6.
(d)
Interpretation:
The curved arrow mechanism for the given reaction is to be stated.
Concept introduction:
The curved-arrow notation is used to show the transfer of electrons from one atom to another. The curved arrow has two barbs (head and tail) which represent the direction of electron flow.
Answer to Problem 19.64AP
The curved arrow mechanism for the given reaction is shown below.
Explanation of Solution
The given reaction is shown below.
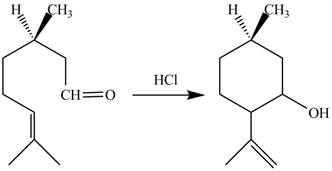
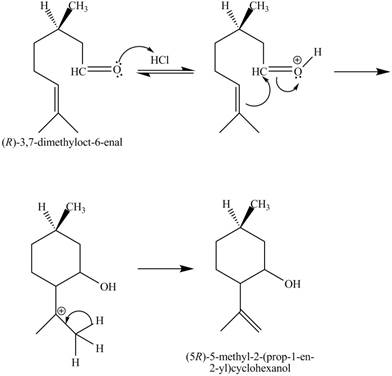
Figure 7
In the above reaction,
The curved arrow mechanim for the given reaction is shown below.
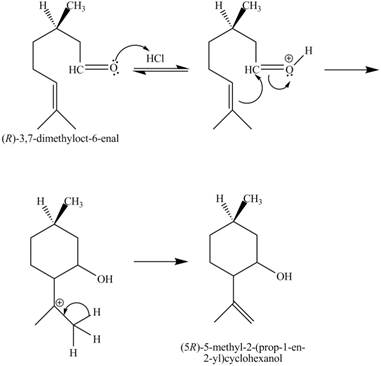
Figure 8
In the above mechanism, the carbonyl carbon of the compound
The curved arrow mechanism of the giveh reaction is shown in Figure 8.
(d)
Interpretation:
The curved arrow mechanism for the given reaction is to be stated.
Concept introduction:
The curved-arrow notation is used to show the transfer of electrons from one atom to another. The curved arrow has two barbs (head and tail) which represent the direction of electron flow.
Answer to Problem 19.64AP
The curved arrow mechanism for the given reaction is shown below.
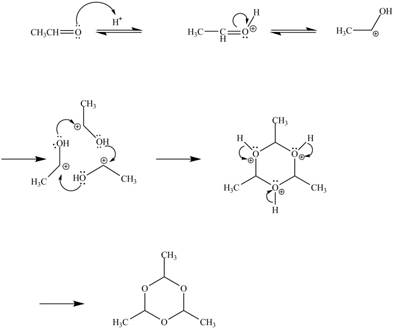
Explanation of Solution
The given reaction is shown below.
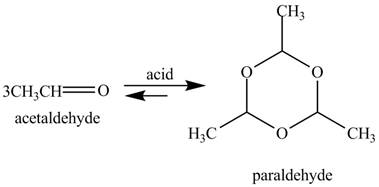
Figure 9
In the above reaction, acetaldehyde in the presence of acid catalyst yields a compound paraldehyde.
The curved arrow mechanism for the given reaction is shown below.
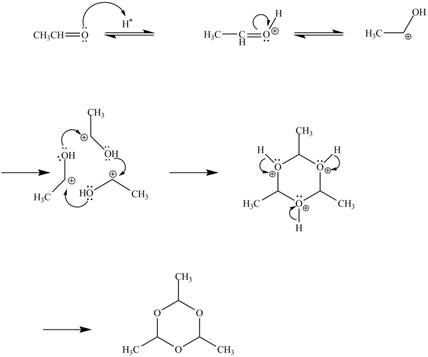
Figure 10
In the above mechanism, the acetaldehyde attacks the proton of acid catalyst to yield protonated acetaldehyde. The three moles of protonated acetaldehyde combines with each other to gives a cyclic structure. This cyclic structure on rearrangement gives the required product paraldehyde.
The curved arrow mechanism of the given reaction is shown in Figure 10.
Want to see more full solutions like this?
Chapter 19 Solutions
Organic Chemistry
- Which compound (i or ii) is the stronger base? Discuss your answer comprehensively by amoungst other providing an acid base reaction for one of the compounds.arrow_forwardWhich compound (i or ii) is the stronger base? Discuss your answer comprehensively by amongst other providing an acid base reaction for one of the compounds.arrow_forwardGive the clear handwritten answer and give the mechanism of given bleow reactionsarrow_forward
- Give the machanism of this reactionarrow_forwardGive the major organic product of the following reaction for a) or b)arrow_forwardCompound X was soluble in water and ether, and its aqueous solution turned litmus blue. It reacted with sodium to give a gas. The compound reacted with benzenesulfonyl chloride and base to give an insoluble product, which was unchanged with acidification. It reacted with nitrous acid to give a yellow solid. Compound A could bearrow_forward
- Compound A was oxidized with periodic acid to give B, which after acid hydrolysis gave C. Bromine oxidation of C gave D. Suggest structural formulas, including stereochemistry, for compounds B, C, and D.arrow_forwardWhat happens when benzene and anisole react with sodium metal in the presence of ethanolic solution of ammonia. Also give mechanism for the reaction of benzene.arrow_forwardGive the structure of a, b, c, d and e in clear handwritten?arrow_forward
- Give a clear handwritten answer...give the mechanism with textual explanation and choose correct option..?arrow_forwardGive the clear handwritten answer...give the mechanism and give the name of missing regent...?arrow_forwardWith diagrams and detailed explanations, comment on the electrophilic aromatic substitution reactivity of quinoline and isoquinolinearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
