
ORGANIC CHEMISTRY MASTERINGCHEM ACCESS
9th Edition
ISBN: 9780137249442
Author: Wade
Publisher: INTER PEAR
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2.51SP
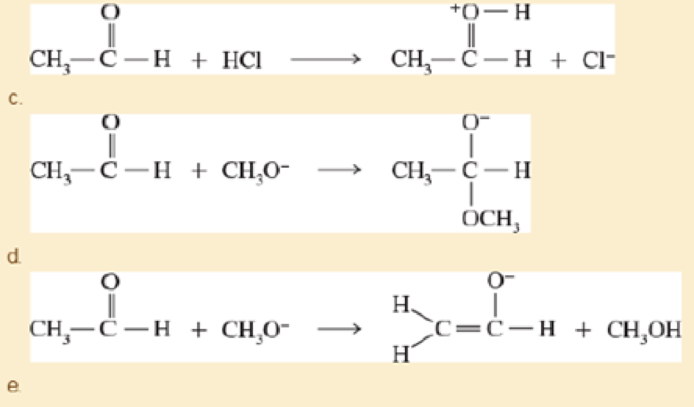
In each reaction, label the reactants as Lewis acids (electrophiles) or Lewis bases (nucleophiles) Use curved arrows to show the movement of electron pairs in the reactions Draw any nonbonding elections to show how they participate in the reactions.
- a. C H3 2NH+HCl → CH3 2N+H2+Cl−
- b. CH3 2NH+CH3Cl → CH3 3N+H+Cl−
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Part I. Write the hydrolytic reaction and equilibrium expression for
the compounds below.
1. Propanoic acid (CH3CH2COOH)
2. Ethylamine (CH;CH2NH2)
2. For each of the following acid-base reactions, predict the products. Determine whether the reaction is favored
as written. Label the acid, base, conjugate acid and conjugate base.
a.
CH₂CH=CH₂
NaNH,
b.
C.
d.
CH₂ONa
NaOH
NaNH,
+
+
+
CH3COOH
H₂
CH3CH2OH
4
Na
reactant.
Write the reaction between ammonia, NH3, and hydrosulfuric acid, H2S.
NH3
+
+
In this reaction
completes the reactant side while
and
complete the products side.
а. NH3
b. NH4+1
с. HS-1
d. H2S
е. Н20
f. H30+1
9. ОН-1
Chapter 2 Solutions
ORGANIC CHEMISTRY MASTERINGCHEM ACCESS
Ch. 2.1A - Prob. 2.1PCh. 2.1B - The NF bond is more polar than the NH bond: but...Ch. 2.1B - For each of the following compounds 1. Draw the...Ch. 2.1B - Two isomers of 1,2-dichloroethene are known One...Ch. 2.2C - Prob. 2.5PCh. 2.2C - Prob. 2.6PCh. 2.3 - Prob. 2.7PCh. 2.4 - Calculate the pH of the following solutions a....Ch. 2.6A - Ammonia appears in Table 2-2 as both an acid and a...Ch. 2.7 - Write equations for the following acid-base...
Ch. 2.7 - Ethanol, methylamine. and acetic acid are all...Ch. 2.8 - Prob. 2.12PCh. 2.10 - Write equations for the following acid-base...Ch. 2.10 - Rank the following acids in decreasing order of...Ch. 2.11 - Prob. 2.15PCh. 2.11 - Prob. 2.16PCh. 2.11 - Consider each pair of bases and explain which one...Ch. 2.12 - Which is a stronger base ethoxide ion or acetate...Ch. 2.12 - Prob. 2.19PCh. 2.12 - Prob. 2.20PCh. 2.12 - Prob. 2.21PCh. 2.12 - Choose the more basic member of each pair of...Ch. 2.14 - Prob. 2.23PCh. 2.15D - Classify the following hydrocarbons and draw a...Ch. 2.16D - Prob. 2.25PCh. 2.17C - Draw a Lewis structure and classify each of the...Ch. 2.17C - Circle the functional groups in the following...Ch. 2 - The CN triple bond in acetonitrile has a dipole...Ch. 2 - Prob. 2.29SPCh. 2 - Sulfur dioxide has a dipole moment of 1.60 D....Ch. 2 - Which of the following pure compounds can form...Ch. 2 - Predict which member of each pair is more soluble...Ch. 2 - Prob. 2.33SPCh. 2 - Prob. 2.34SPCh. 2 - Predict which compound in each pair has the higher...Ch. 2 - All of the following compounds can react as acids...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - The Ka of phenylacetic acid is 5 2 105, and the...Ch. 2 - The following compound can become protonated on...Ch. 2 - The following compounds are listed in increasing...Ch. 2 - Prob. 2.42SPCh. 2 - Prob. 2.43SPCh. 2 - Compare the relative acidity of 1-molar aqueous...Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - The following compounds can all react as bases. a....Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - Prob. 2.48SPCh. 2 - Methyllithium (CH3Li) is often used as a base in...Ch. 2 - Label the reactants in these acid-base reactions...Ch. 2 - In each reaction, label the reactants as Lewis...Ch. 2 - Prob. 2.52SPCh. 2 - Each of these compounds can react as a nucleophile...Ch. 2 - Prob. 2.54SPCh. 2 - Give a definition and an example for each class of...Ch. 2 - Circle the functional groups in the following...Ch. 2 - Prob. 2.57SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 19. Choose the correct reagents/reactants to synthesize the following compound. 1. NaH 1. NaH A. OH 2. OH 2. Br Br 3. H3O* С. 1. NaH 1. NaH HO 2. HO 2. Br Br 3. H;0* B. D.arrow_forwardComplete the reactions below, briefly explaining in each case type of reaction taking place. i. Δ N-Ph ??? 1. Br2, NaOH ii. ??? 2. H+, H₂O + CHBг3 OHarrow_forwardWater will be formed in the following reaction. Write the other product that will be formed. HF + KOH ⟶⟶ H2O + ____arrow_forward
- Consider the acid-base reaction below: (Question 14) Ca(HCO3)2 + Ca(OH)2 ----> Compound A + H2O (OR Ca(HCO3)2 + Ca left parenthesis OH right parenthesis 2 ----> Compound A + H2O) One of the single ions that is used to form compound A is ____. Assume enough of each compound is used to get a complete reaction. Question 14 answer choice options: C32- (OR C3 2 minus) C43- (OR C4 3 minus) C3- (OR C 3 minus) CO- (OR C O 1 minus) CO2- (OR C O 2 minus) CO43- (OR C O4 3 minus) CO4- (OR C O4 1 minus) C22- (OR C2 2 minus) CO32- (OR C O3 2 minus) C4- (OR C4 1 minus) C2- (OR C 2 minus) C33- (OR C3 3 minus) CO6- (OR C O 6 minus) C23- (OR C2 3 minus) C2- (OR C2 1 minus) C- (OR C 1 minus) C3- (OR C3 1 minus)…arrow_forwardComplete the reactions aa CI CI НО OH НО. позвон досв НО. OH НО OH OH НО H₂N _OH _OH NH₂arrow_forward1. Complete and balance the following neutralization equations. H2SO4 + NaOH -> __ +__ ____+_____ -> BaCl2 + H2O ____+ KOH -> K3PO4 + ____ 2. How is a neutralization reaction related to a saponification reaction?arrow_forward
- Explain why the pH of 0.1 M ethanol is higher than the pH of 0.1 M acetic acidarrow_forward6. Draw the product of the proton transfer reaction. Label the acid and base in the starting materials, and the conjugate acid and base in the products. OH + CH3ONaarrow_forward7. Use the curved arrow notations to show the flow of electrons in the given reactions. 4. a. b. OH OH 4. → OH. ofarrow_forward
- 1. Write the equation for the hydrolysis of aniline (C6H5NH2), an organic base that smells like rotten fish. 2. Determine whether the following conjugates can undergo hydrolysis. Type either YES or NO. If it can hydrolyze, write the hydrolysis equation. A. ClO4- B. CH3NH3+arrow_forwardSelect all compounds that are able to deprotonate ethanol, EtOH, to the extent that the neutralization reaction is product-favored at equilibrium. a. Na2s b. NaNH2 С. BuLi d. NaF e. NaCN f. NaCH3CO2 g. NH3 h. CH3LIarrow_forwardWhat is the product of the reaction? EtO Na* ELOH ?? HO. O`Na+ OEt HO. OEt OEtarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY