
Concept explainers
(a)
Interpretation:
The formal charge on each atom has to be determined and structure of lower energy has to be identified.

Concept Introduction:
The formal charge on each atom in the Lewis structure can be calculated from the equation written as follows:
Here,
Lowest energy structure is the one that has zero or nearly zero formal charge.
(a)
Explanation of Solution
The formal charge on each atom in the Lewis structure can be calculated from the equation as follows:
For structure 1:
Substitute 5 for
Substitute 4 for
Similarly for other Lewis structure, substitute 5 for
Substitute 5 for
Substitute 4 for
Hence formal charge on each structure is illustrated below:

Since the former has less formal charges on maximum number of atoms so I is lower in energy than II therefore I represents structure of lowest energy.
(b)
Interpretation:
The formal charge on each atom has to be determined and structure of lower energy has to be identified.
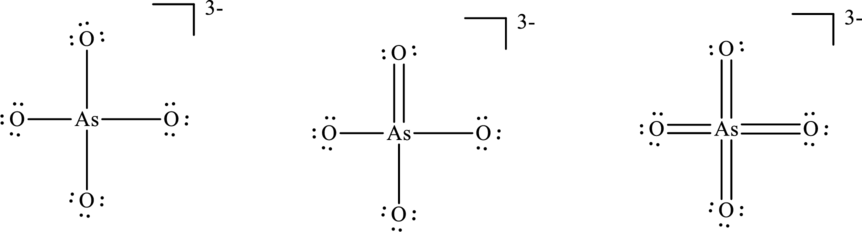
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
The formal charge on each atom in the Lewis structure can be calculated from the equation as follows:
For structure I:
Substitute 6 for
Substitute 6 for
Substitute 5 for
For structure II:
Similarly for second Lewis structure, substitute 6 for
Substitute 6 for
Substitute 5 for
For structure III:
Similarly for third Lewis structure, substitute 6 for
Substitute 5 for
Hence formal charge in the two structures is illustrated below.
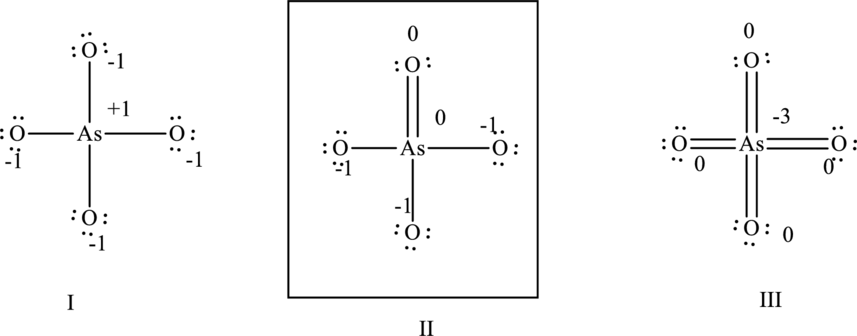
Since the II has a
(b)
Interpretation:
The formal charge on each atom has to be determined and structure of lower energy has to be identified.
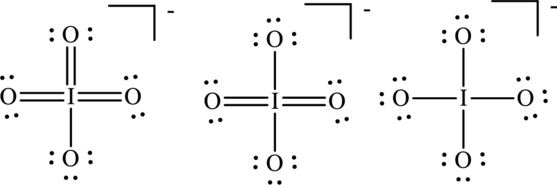
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
The formal charge on each atom in the Lewis structure can be calculated from the equation as follows:
For Lewis structureI, substitute 6 for
Substitute 6 for
Substitute 7 for
For Lewis structure II,
Substitute 6 for
Substitute 6 for
Substitute 7 for
For Lewis structure III, substitute 6 for
Substitute 6 for
Substitute 7 for
Hence formal charge in the three structures is illustrated below:
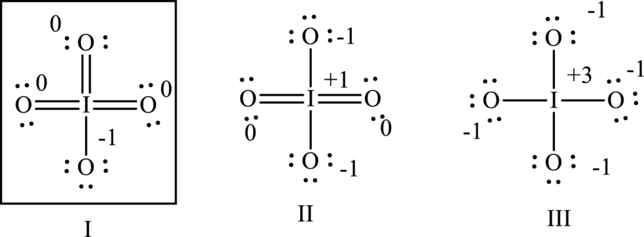
Since I has zero formal charges on central electronegative iodine atom compared to other cases with
Want to see more full solutions like this?
Chapter 2 Solutions
EBK CHEMICAL PRINCIPLES
- the formal charges on all the atoms in the following Lewis diagrams. Which one would best represent bonding in the molecule Cl2O ?arrow_forwardDo any of the atoms in an ibuprofen molecule have a nonzero formal charge?arrow_forwardAssign formal charges to all atoms in the following Lewis diagrams. (a) SO42 (b) S2O32 (c) SbF3 (d) SCNarrow_forward
- A complete Lewis structure must show all nonzero formal charges. Complete each of thefollowing Lewis structures by adding any missing formal charges.arrow_forwardThree known isomers exist of N2CO, with the atoms in these sequences: NOCN; ONNC; and ONCN. Write resonance structures for each isomer and use formal charge to predict which isomer is the most stable.arrow_forwardWrite the Lewis structure and chemical formula of the compound with a molar mass of about 70 g/mol that contains 19.7% nitrogen and 80.3% fluorine by mass, and determine the formal charge of the atoms in this compound.arrow_forward
- Write all possible resonance structures for the following species. Assign a formal charge to each atom. In each case, which resonance structure is the most important? (a) NO2 (nitrogen is central) (b) ClCNarrow_forwardCarbon monoxide (CO) is an example of an overall neutral molecule (netcharge=0) that hasnon-zero formal charges. Draw a Lewis structure of carbon monoxide (CO).arrow_forwardTwo Lewis structures can be written for nitrosyl fluoride, which contains one nitrogen, one oxygen, and one fluorine atom per molecule. Write the two Lewis structures land assign a formal charge to each atom.arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





