
Concept explainers
Interpretation: On addition of one mole of
Concept Introduction:
Addition reaction of dienes:
The diene with a four carbon system is
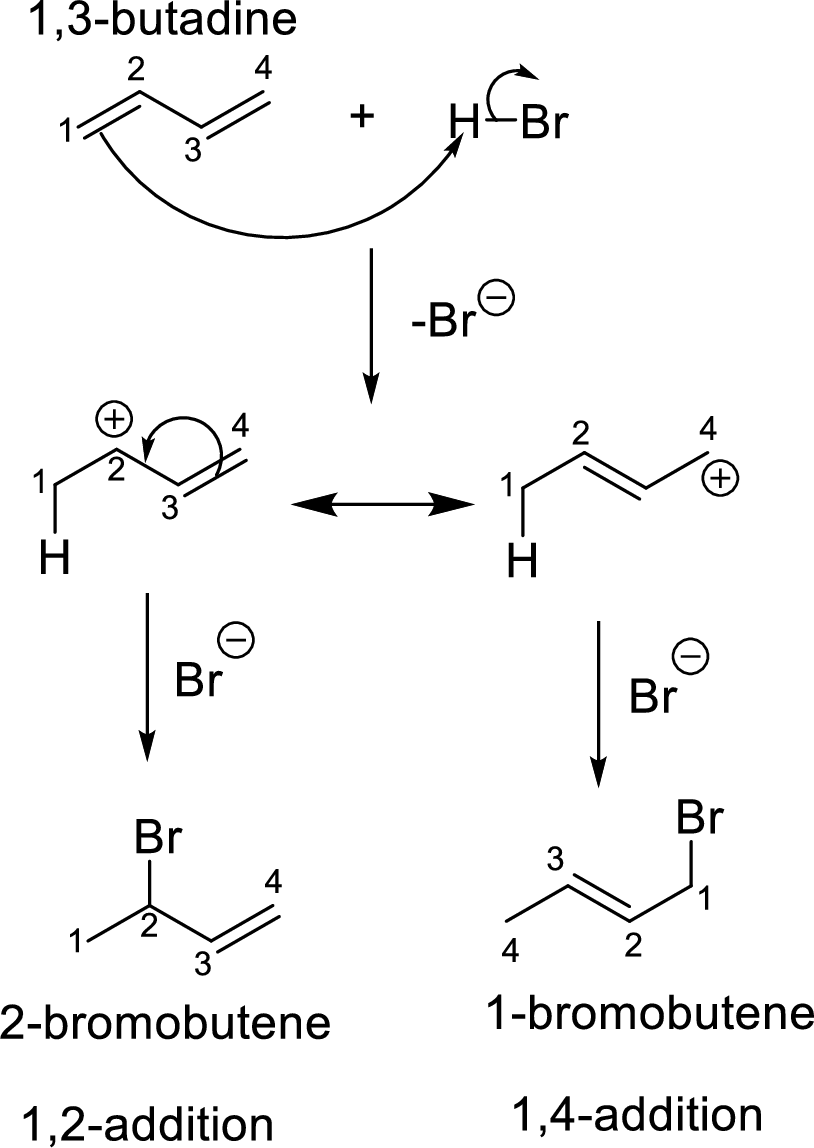
The constitutional isomers have same molecular formula and different connectivity of the substituents in the same structural formula.
Example:
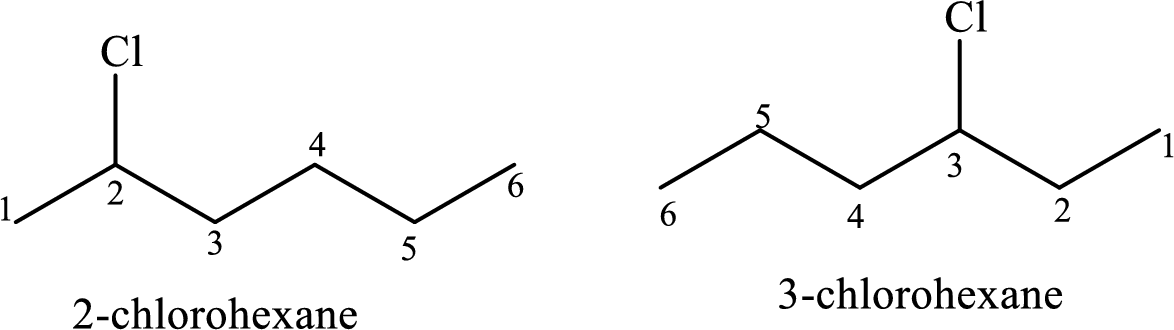
Trending nowThis is a popular solution!

Chapter 20 Solutions
Organic Chemistry
- draw structures for all the isolated, conjugated, and cumulated dienes of molecular formula C5H8.arrow_forwardAccount for the fact that addition of HCl to 1-bromopropene gives exclusively 1-bromo-1-chloropropane.arrow_forwardPredict the relative stabilities of alkenes and cycloalkenes based on their structure and stereochemistry.arrow_forward
- Draw the structure(s) of all of the alkene isomers, C5H10, that contain a branched chain. Consider E/Z stereochemistry of alkenes.arrow_forwardWhich will be more stable, cis or trans-1,4-tert-butylcyclohexane? Explain by drawing their structures?arrow_forwardBromine adds to cis- and trans-2-butene to give different diastereomers of 2,3-dibromobutane. What does this say about the mode of addition of bromine to thisalkene?arrow_forward
- Draw a structural formula for the alkene with the molecular formula C5H10 that reacts with Br2 to give each product.arrow_forwardWhich of the statement is INCORRECT? a. The increase in stability of 2,4-hexadiene over 1,3-hexadiene is due to the increased double bond substitution of the former. b. The stabilization of dienes by conjugation is less pronounced than the aromatic stabilization of benzene. c. Resonance description in alkenes usually involves charge separation. d. Higher energy pi-orbitals often have decreasing number of nodes.arrow_forwardwrite structural formulas for all the compounds that are trichloro derivatives of cyclopropane, which are chiral and which are achiral?arrow_forward
- what are the physical porpeties of 1,3,5-cyclohexene thank youarrow_forwardReaction of HBr with 3-methylcyclohexene yields a mixture of four products: cis-and trans-1-bromo-3-methylcyclohexane and cis-and trans-1-bromo-2-methylcyclohexane. The analogous reaction of HBr with 3-bromocyclohexene yields trans-1, 2-dibromocyclohexane as the soleproduct. Draw structures of the possible intermediates, and then explain why only a single product is formed in this reaction.arrow_forwardThe major product of the following reaction exists as two stereoisomers. Draw both isomers: show all hydrogen atoms in the structures. Use wedge and dash bonds to indicate the stereochemistry (you don’t need to draw wedge and dash bonds for C-H bonds of CH3 and CH2 groups). Assign stereo configuration of the asymmetric carbon atoms and write the relationship between two isomers.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
