
Concept explainers
(a)
Interpretation: The important resonance hybrids for the given allylic carbocations have to be drawn and ranked based on their contributions.
Concept Introduction:
Allylic systems:
The allylic system is characterized by the connectivity of a single bonded carbon atom connected to double bond
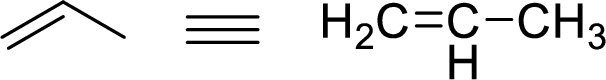
Stability of the allylic carbocations:
Example for Primary allylic carbocation is:
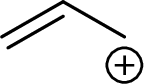
Example for Secondary allylic carbocation is:
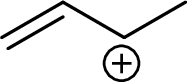
Example for tertiary allylic carbocation is:
(b)
Interpretation: The important resonance hybrids for the given allylic carbocations have to be drawn and ranked based on their contributions.
Concept Introduction:
Allylic systems:
The allylic system is characterized by the connectivity of a single bonded carbon atom connected to double bond alkene functional group as shown here:
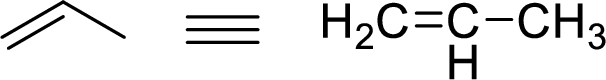
Stability of the allylic carbocations:
Example for Primary allylic carbocation is:
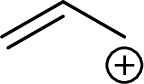
Example for Secondary allylic carbocation is:
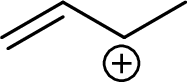
Example for tertiary allylic carbocation is:
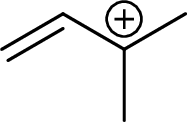
(c)
Interpretation: The important resonance hybrids for the given allylic carbocations have to be drawn and ranked based on their contributions.
Concept Introduction:
Allylic systems:
The allylic system is characterized by the connectivity of a single bonded carbon atom connected to double bond alkene functional group as shown here:
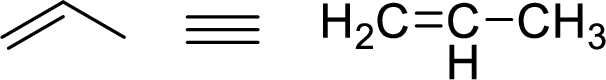
Stability of the allylic carbocations:
Example for Primary allylic carbocation is:
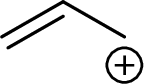
Example for Secondary allylic carbocation is:
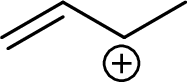
Example for tertiary allylic carbocation is:
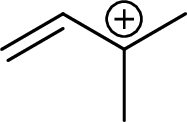
Trending nowThis is a popular solution!

Chapter 20 Solutions
Organic Chemistry
- Give a clear explanation of how the followingcarbocation is stabilized:arrow_forwardRank the followings carbocations in terms of increasing stability (least to most)arrow_forwardPlease provide a curved-arrow mechanism for the following reaction. Be sure to use your mechanism and a few words to explain the regiochemistry of the bromines in the final product and why each carbocation intermediate in your mechanism is the preferred intermediate for that step. Include ALL lone pairs and formal charges. Thank you.arrow_forward
- For each reaction shown, determine which substitution mechanism (SN1 or SN2) is likeliest to predominate, and provide two reasons to support your answer.arrow_forwardWrite an additional resonance contributing structure for each carbocation and state which of the two makes the greater contribution to the resonance hybrid. Classify each additional contributing structure as a 1°, 2°, or 3° allylic cationarrow_forwardRank the compounds in each set from most to least reactive in an EAS reactionarrow_forward
- Rank the following alkenes in order of increasing stability (least to most stable)arrow_forwardThe resonance-stabilized carbocation formed in step one has two electrophilic centers. Draw the final step of the mechanism using both provided resonance structures. arrow_forwardRank the following carbanions in order of increasing stability:arrow_forward
- This is a Diels-Alder reaction between furan + maleic anhydride (endo and exo products). For each cycloaddition product, draw in all hydrogen atoms, and write the molecular formula below.arrow_forwardThe following compound readily eliminates CO; to form a conjugated six membered ring. (a) Complete the reaction with drawing the possible structure and (b) explain why this reaction goes steadily.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

