
Interpretation:
Whether an achiral or two equal enantiomeric products would be given has to be predicted in the product of the given reaction and explained with chair like transition state.
Concept Introduction:
Cope-rearrangement:
It is a pericyclic reaction that involves the redistribution of six electrons through the formation of a cyclic transition state from which a
Example with mechanism of cope-arrangement:
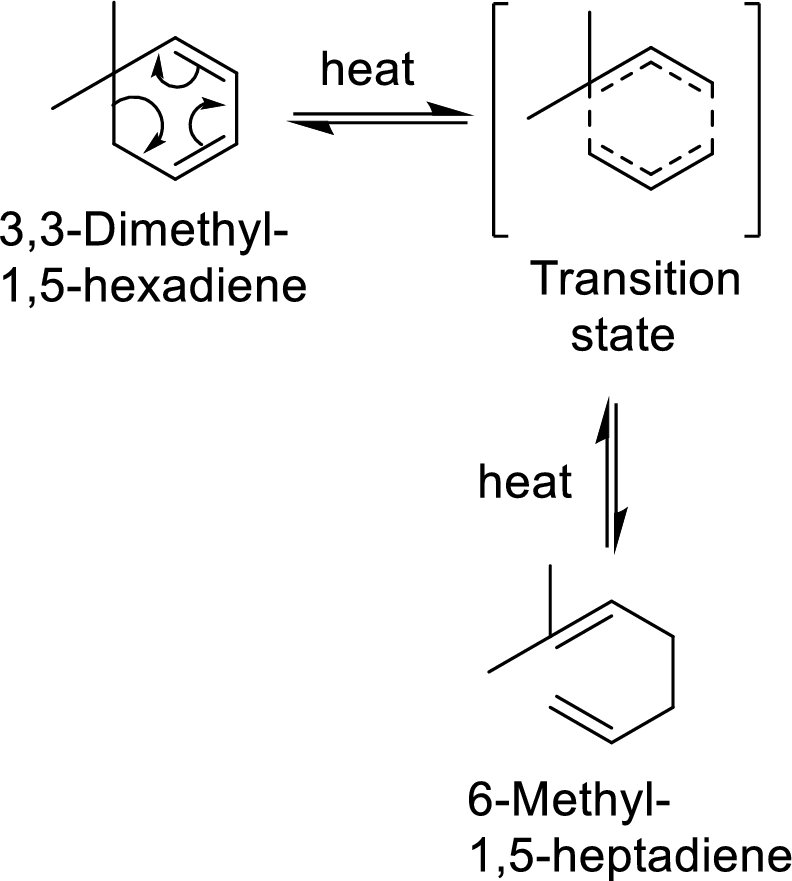
In this mechanism, two pi-bonds and one sigma bond of the reactant molecule has been rearranged and formed two new pi-bonds through a cyclic transition state.
Identification of cope-rearrangement in a
In the cope-rearrangement, the flow of electrons takes place between six bonds that are bonded as
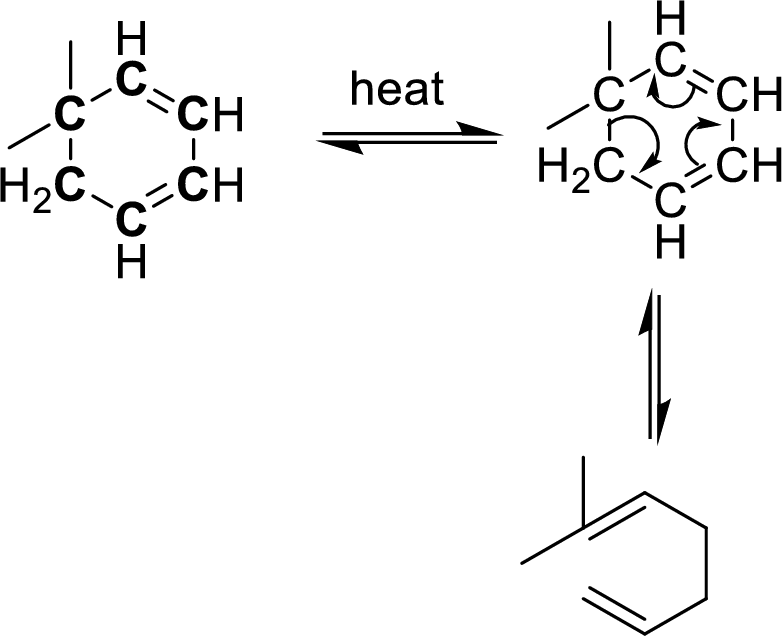
The carbon atoms that are involving in the cope-rearrangement are shown in bold.
Stereochemistry in a product formed:
- • In the product of a
chemical reaction , if a carbon atom has been attached with four different carbon atoms, then it is known as chiral carbon atom or stereocenter in the product. - • The bonds of the functional groups because of which a new chiral carbon is supposed to form have to be represented in solid wedge bond and hashed-wedge bonds according to the particular enantiomer.
- • Racemic mixture is the mixture of two enantiomers in equal proportions.
- • Enantiomers are non-superimposable mirror images.
- • Achiral product is the product in which there won’t be any chiral centre or stereocenter.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Organic Chemistry
- Consider the substitution reaction that takes place when (R)-3-bromo-3-methylhexane is treated with sodium methoxide. Deduce the mechanism of this reaction working step by step. Write all possible products showing the stereochemistry.arrow_forwardDraw the products of the SN1 reaction below and use wedge-and-dash bonds to indicate the stereochemistry of any stereogenic centers.arrow_forwardif you start with a chiral alkyl halide in your synthesis of an alcohol and you perform an SN2 type of reaction would you expect inversion of the configuration or a racemic mixture of a product?arrow_forward
- Which reaction mechanism will create a shift in the stereochemistry of the resulting product? a SN1 b SN2 c E1 d E2arrow_forwardThe following reaction proceeds through an E2 mechanism with two potential isomers as products. Which do you expect to the major product? Explain your answer using Newman projections.arrow_forwardConformation control of reaction mechansims. Consider the reaction below: Provide all possible elimination products from this reaction and identify the major elimination product. Provide mechanisms (with curved arrows) that clearly explain the reaction outcome.arrow_forward
- Optically active butan-2-ol racemizes in dilute acid. Propose a mechanism for this racemization.arrow_forwardThe following chiral compound undergoes a stereospecific E2 reaction, where only one stereoisomer is produced as product. What is the product of the reaction?arrow_forwardWhen the compound shown below undergoes acid-catalyzed dehydration, a ring expansionoccurs to give the fused-ring product. What type of intermediate is formed in this reaction?Explain the ring expansion in terms of the reaction mechanismarrow_forward
- When cyclopentene is treated with Br2 in the presence of CH2Cl2 solvent, the major substrate product is __ .arrow_forwardWhen the given alkene undergoes hydroboration followed by oxidation, which would be produced in regards to the products stereochemistry a. A mix of diastereomers b. A single enantiomer c. A racemic mixture d. An achiral productarrow_forwardConsider diastereomers X and Y shown below. Cl diastereomer X Cl HO H OH H Each diastereomer, if treated with sodium ethoxide (NaOCH2CH3), undergoes an E2 reaction. One of the diastereomers undergoes an E2 to make a single compound. The other diastereomers undergoes E2 eliminations leading to two different compounds. Draw the mechanisms for all three of these E2 reactions, being sure to use conformational structures for the substrate in each reaction. Which of the two diastereomers will undergo an E2 reaction at the faster rate? Why? (In organic chemistry it is often said one picture is worth a thousand words. Be sure to include a picture that supports whatever argument you are making.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

