
Concept explainers
(a)
Interpretation: The mechanism for the formation of compound A has to be proposed.
Concept Introduction:
Diels-Alder reaction:
It is the reaction of conjugated dienes with double or triple bonded compounds which are known as “dienophiles”. The reaction is a
Example:
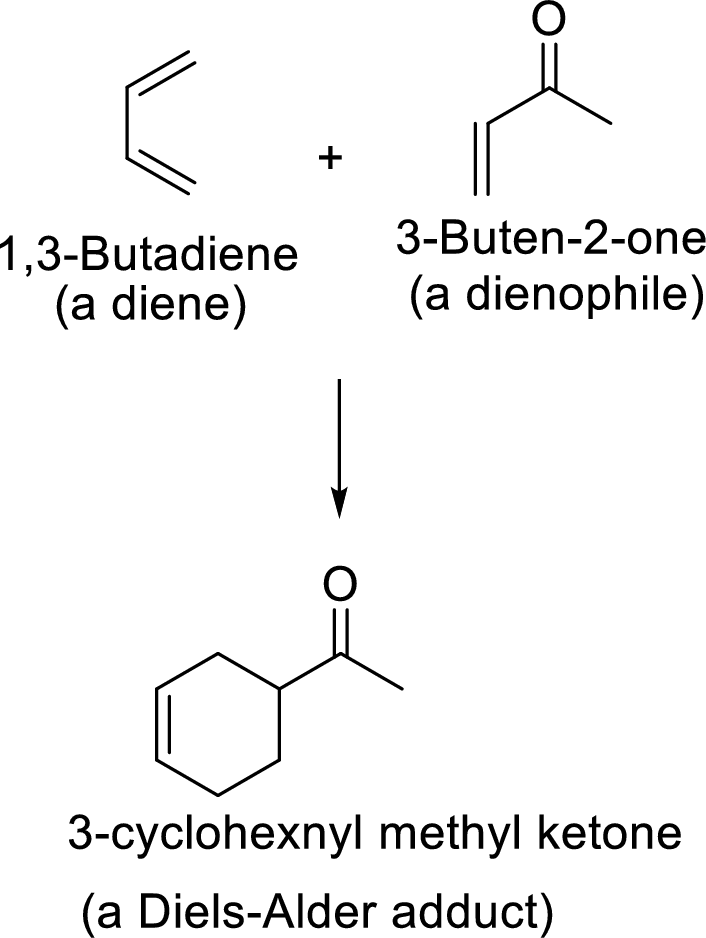
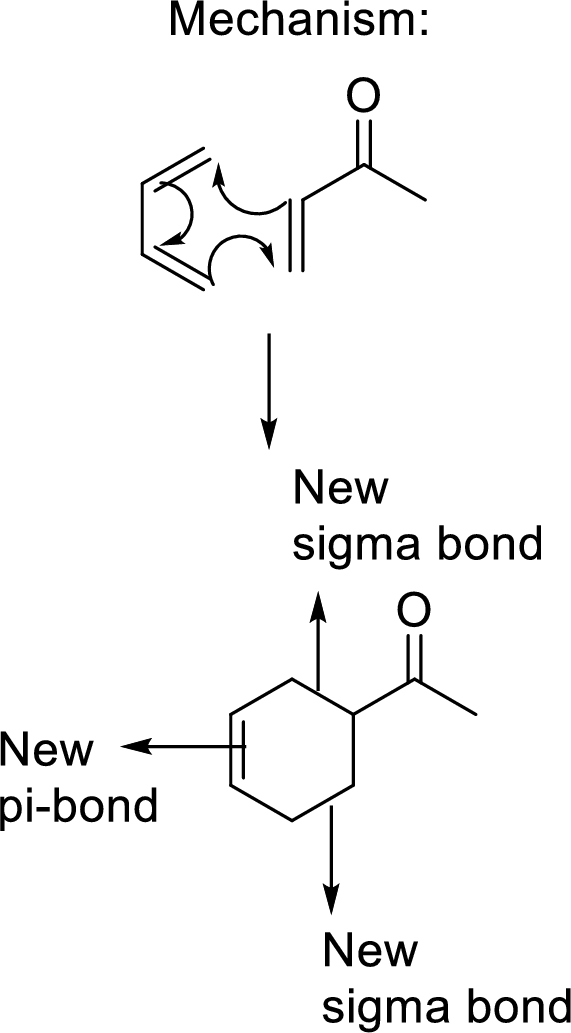
This mechanism shown that three
Formation of Benzyne:
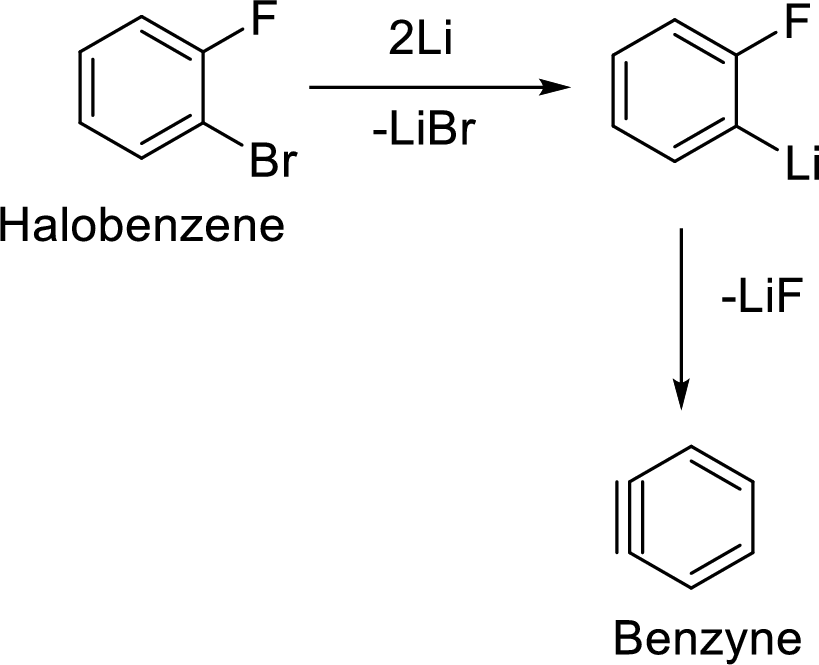
The elimination of two halogens from the halobenzene forms the most reactive neutral intermediate with triple bond which is known as Benzyne intermediate.
Diels-Alder reaction with Benzyne intermediate:
The reaction of Benzyne with furan serves as an example for this reaction:
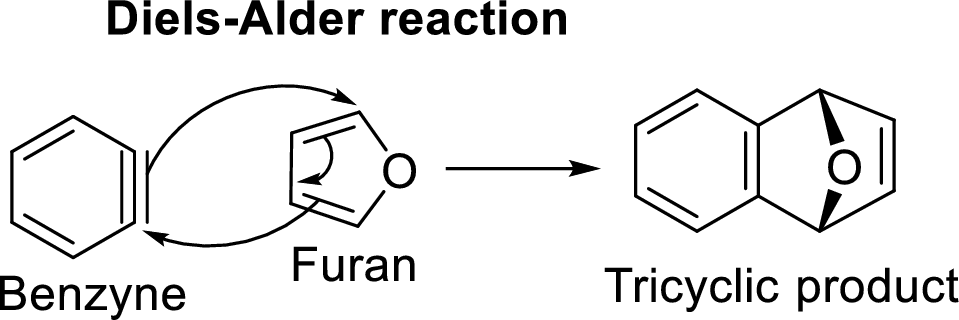
Benzyne is an extremely reactive species because of the presence of triple bond in it. It undergoes Diels-Alder reaction in which it acts as dienophile and reacts with furan which acts a diene. The resulted product will be a tricyclic product which is known as Diels-Alder adduct.
(b)
Interpretation: Using the given sources, the conversion of compound (A) into Tolciclate has to be shown.
Concept Introduction:
Acidic hydrolysis of ether:
Ether on hydrolysis gets converted into two moles of alcohols of same type in the case of symmetrical ether and of different types in the case of unsymmetrical ether. .
General scheme for symmetrical ether:
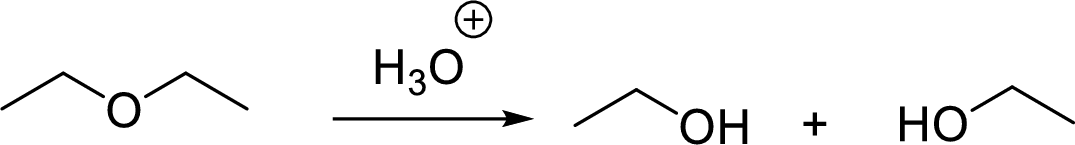
General scheme for unsymmetrical ether:
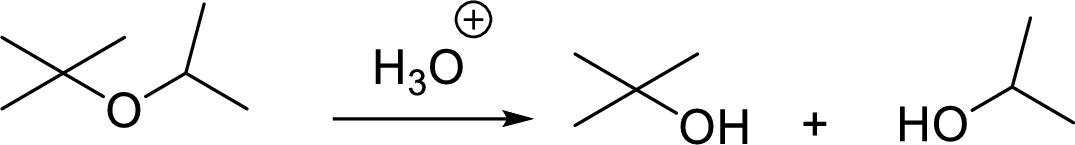
Tosylation reaction:
When an alcohol is treated with any tosyl chloride (methane sulfonyl chloride) it gets converted into tosylated product and this reaction is called as Tosylation reaction which is shown below:
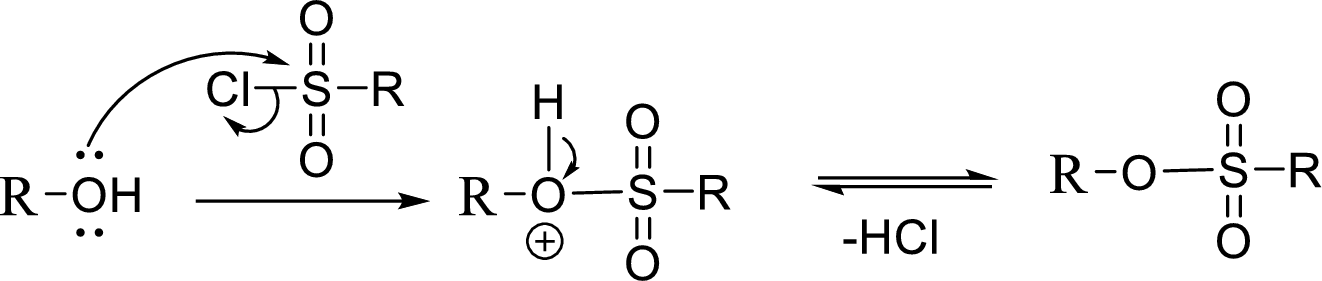
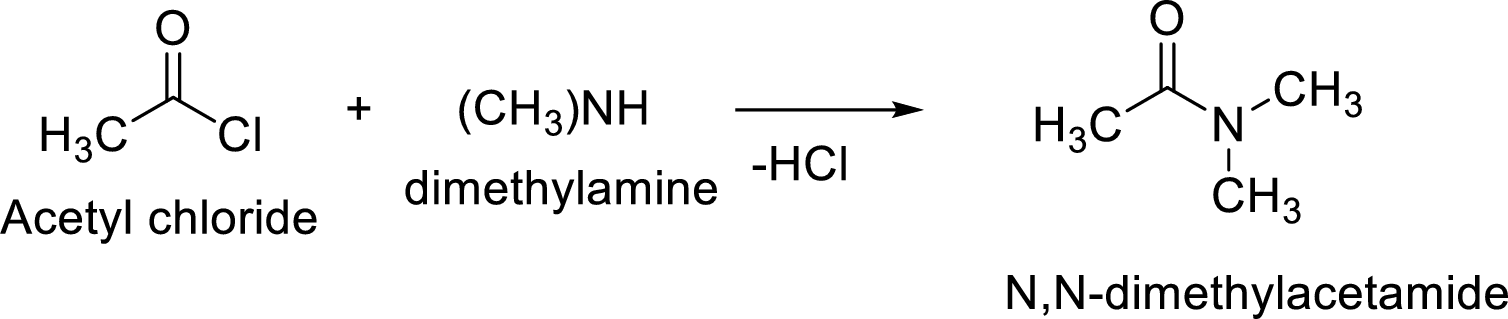
The reaction of acetyl chloride with
The acyl group of acetyl chloride is very reactive and the chloride group attached to it is a good leaving group. The nucleophile from amine attacks the carbonyl carbon of the acetylchloride to form N- methylacetamide. The chloride group leaves and gets attached to hydrogen of the amine and leaves as hydrochloric acid.
The reaction is:
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Organic Chemistry
- One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.arrow_forwardTreatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.arrow_forwardFollowing is a retrosynthetic analysis for an intermediate in the industrial synthesis of vitamin A. (a) Addition of one mole of HCl to isoprene gives 4-chloro-2-methyl-2-butene as the major product. Propose a mechanism for this addition and account for its regioselectivity. (b) Propose a synthesis of the vitamin A precursor from this allylic chloride and ethyl acetoacetate.arrow_forward
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardCompound A (C9H18O) forms a phenylhydrazone, but it gives a negative Tollens’ test. The IR spectrum of A has a strong band near 1710 cm−1 . The broadband carbon NMR spectrum of A is given below. a)Propose a structure for A and explain how your structure fits the spectral data. b) Propose a mechanism for this reaction.arrow_forwardShow the products you would obtain by acid-catalyzed reaction of cyclohexanone with ethylamine, CH3CH2NH2, and with diethylamine, (CH3CH2) 2NH.arrow_forward
- Azlactones are important starting materials used in the synthesis of dehydro α-aminoacids. They react with aldehydes to form an intermediate that is hydrolyzed under acidic conditions to give the final amino acid product. Provide the structure of the intermediate and propose a mechanism for its formation.arrow_forwardCompound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forwardA step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


