
Interpretation:
The products in the given cope rearrangements has to be predicted with stereochemistry.
Concept Introduction:
Cope-rearrangement:
It is a pericyclic reaction that involves the redistribution of six electrons through the formation of a cyclic transition state from which a
Example with mechanism of cope-arrangement:
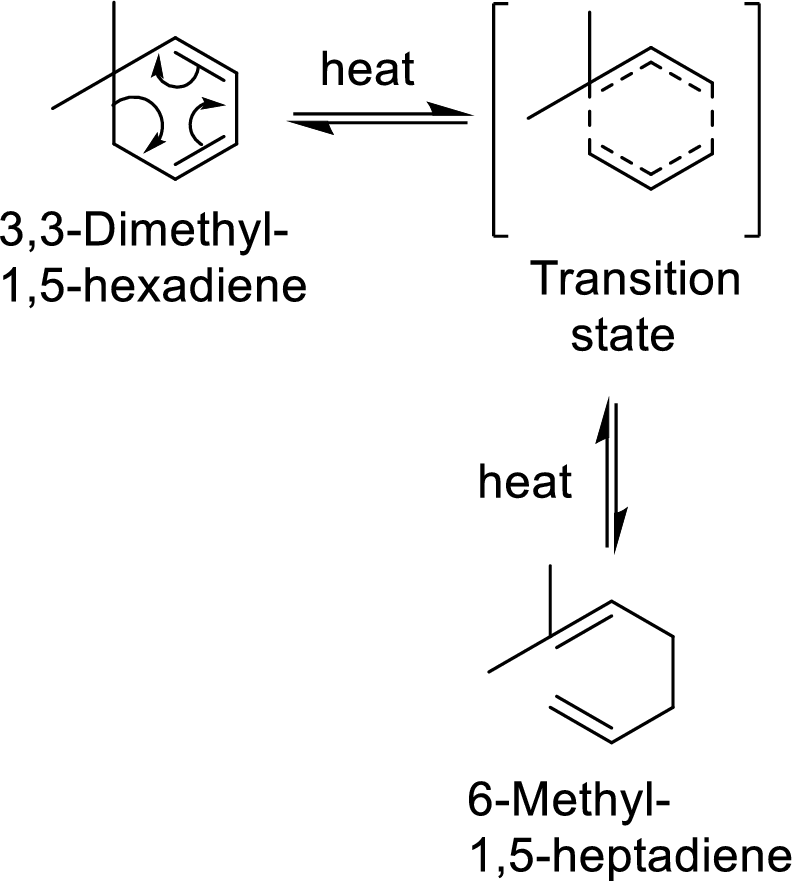
In this mechanism, two pi-bonds and one sigma bond of the reactant molecule has been rearranged and formed two new pi-bonds through a cyclic transition state.
Identification of cope-rearrangement in a
In the cope-rearrangement, the flow of electrons takes place between six bonds that are bonded as
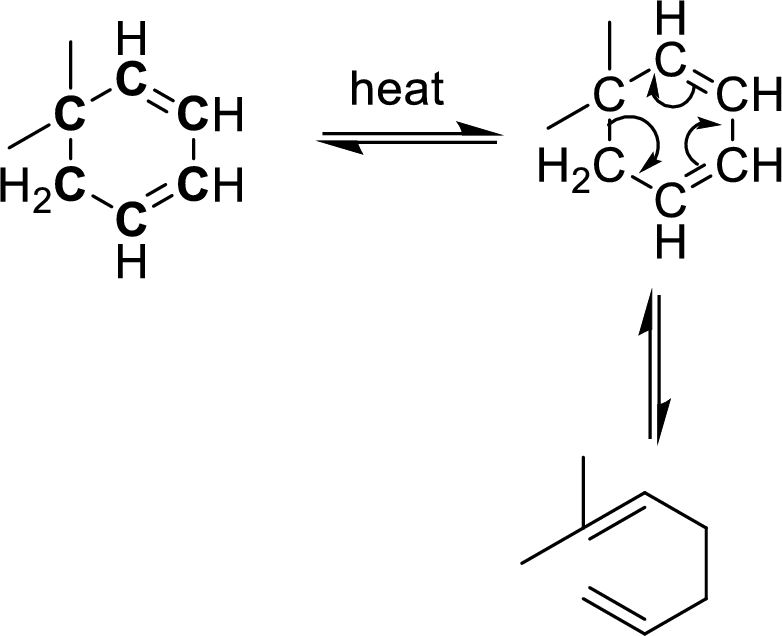
The carbon atoms that are involving in the cope-rearrangement are shown in bold.
Stereochemistry in a product formed:
- • In the product of a
chemical reaction , if a carbon atom has been attached with four different carbon atoms, then it is known as chiral carbon atom or stereocenter in the product. - • The bonds of the functional groups because of which a new chiral carbon is supposed to form have to be represented in solid wedge bond and hashed-wedge bonds according to the particular enantiomer.
- • Racemic mixture is the mixture of two enantiomers in equal proportions.
- • Enantiomers are non-superimposable mirror images.
- • Achiral product is the product in which there won’t be any chiral centre or stereocenter.
Trending nowThis is a popular solution!

Chapter 20 Solutions
Organic Chemistry
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forwardThe aryl diene undergoes sequential Heck reactions to give a product with the molecular formula C15H18. Propose a structural formula for this product.arrow_forward
- It is not uncommon for organic chemists to prepare acetals by an exchange-type process known as transacetalization. Predict the product(s) and show the mechanism for the transacetalization reactions below.arrow_forwardPredict the product of the following epoxide ring opening and provide a mechanism for its formation. Be sure to include stereochemistry.arrow_forwardOn treatment with HBr, vinylcyclohexane undergoes addition and rearrangement to yield 1-bromo-l-ethylcyclohexane. Using curved arrows, propose a mechanism to account for this result.arrow_forward
- Propose a mechanism to account for the product of the following reactions. Please show the structures of the intermediates and using curved arrows to indicate electron flow in each step. Rearrangements occur.arrow_forwardWrite four examples of the condensation reactions of carbonyl compounds, starting with benzaldehyde (C6H5CHO), with appropriate reagents and appropriate reactions. Write down the mechanism of one you want in detail.arrow_forwardPredict the products for each of the following reactions and propose a mechanism that explains the formation of each product.arrow_forward
- 2,3-Dimethylbutane reacts with bromine in the presence of light to give a mono brominated product. The further reaction gives a good yield of a dibrominated product. Predict the structures of these products, and propose a mechanism for the formation of the mono brominated product.arrow_forwardIn a strongly acidic solution, cyclohexa-1,4-diene tautomerizes to cyclohexa-1,3-diene.Propose a mechanism for this rearrangement, and explain why it is energetically favorablearrow_forwardIntroduce the reaction and structure of the expected products (indicate the main product, when possible) for a reaction of 3-ethyl-5-methyl-hex-2-ene with the following reagents: (a) Br2, CH2Cl2 (b) H2O , Diluted H2SO4.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

