
Interpretation:
The following mechanism, which has the curved arrow omitted, is for another reaction that converts an amide to a carboxylic acid. The mechanism is to be completed by drawing the missing curved arrows, and tetrahedral intermediate is to be identified. Below each reaction arrow, the name of the elementary step that takes place is to be written, and it is to be indicated whether the step is reversible or irreversible.
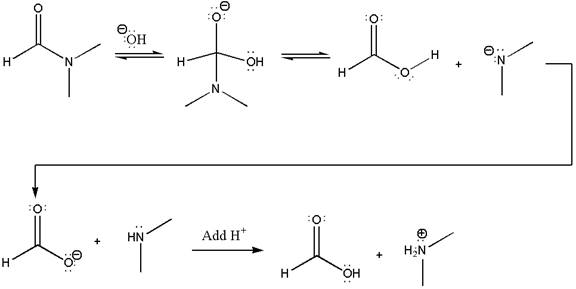
Concept introduction:
The opposite charges attract while the like charges repel. In an elementary step, electrons tend to follow from an electron rich site to an electron poor site. An elementary step can also occur in which only a single bond is broken and both electrons from that bond end up on one of the atoms initially involved in the bond.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- Complete the mechanism for the decarboxylation given by adding any missing atoms, bonds, charges, nonbonding electrons, and curved arrows.arrow_forwardDraw the mechanism and any intermediates for the following reaction. Show the product.arrow_forwardUsing the reaction shown in Box (the attack of an amine on the carbonyl group of a ketone) as a starting point, draw curved arrows to represent the base-catalyzed reaction (when the group —B: is present).arrow_forward
- Complete the following reversible reaction and draw the mechanism of formation ofproducts. Indicate using the arrows of unequal length whether the shift in equilibrium will favor the reactants or the products. give 2 reasonsarrow_forwardCan you help me draw the mechanism arrows for this reaction and determine if the reaction is going forward or reverse?arrow_forwardBy following the curved red arrows, draw the product(s) of each of the following reaction steps:arrow_forward
- Draw the reaction mechanism of the following reaction in acidic conditions. List allpossible compounds in the reaction mixture right after you stop the reactionarrow_forwardUsing the attached image, fill in mechanism arrows for each step of the mechanism. Make sure that your arrows start and end in the correct place. *Acid chlorides have an incredible ability to be substituted into almost any carbonyl functional group. This is due to the fact that chloride makes for an excellent leaving group.arrow_forwardIn a Wittig reaction, a ketone or aldehyde reacts as an electrophile with a nucleophile called a Wittig reagent (or phosphonium ylide) to produce an alkene. The Wittig reagent is commonly synthesized first in a two-step process beginning with an alkyl halide, then reacted with the carbonyl compound. In this problem, you'll explore the mechanism of a multi-step synthesis to make an alkene using the Wittig approach.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
