
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
8th Edition
ISBN: 8220102895805
Author: Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20.3, Problem 20.7P
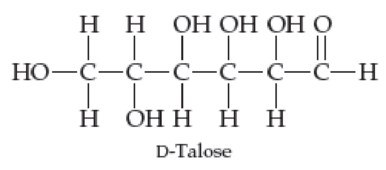
D-Talose, a constituent of certain antibiotics, has the open-chain structure shown next. Draw D-talose in its cyclic hemiacetal form.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The compound shown here is used to treat trypanosome infection. What amino acid does the compound resemble?
A decapeptide which could resist burns was isolated from an endemic plant from Mt. Mayon. Deduce the amino acid sequence of the decapeptide given the following informations.
Mercaptoethanol gave two pentapeptides F1 and F2.
Chymotrypsin treatment of F1 releases S and a tetrapeptide (C,K,F,M) while F2 gave a dipeptide (M,S) and a tripeptide (C,F,K)
F1 when treated with CNBr released a free homoserine lactone and a tetrapeptide.
Trypsin treatment of F1 gave a tripeptide and a dipeptide.
F2 treated with carboxypeptidase released free M while with trypsin gave free K and a tetrapeptide.
How does the structure of chymotrypsin enable it to cleave only peptide
bonds that are adjacent to aromatic R groups? Suppose a protease could only
cleave adjacent to Lysine and Arginine; how would the structure of this protein
differ from that of chymotrypsin?
Chapter 20 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
Ch. 20.1 - Classify the following monosaccharides as an...Ch. 20.1 - Prob. 20.2PCh. 20.2 - Prob. 20.3PCh. 20.2 - Prob. 20.4PCh. 20.2 - Prob. 20.6PCh. 20.3 - D-Talose, a constituent of certain antibiotics,...Ch. 20.3 - Prob. 20.8PCh. 20.3 - Draw the structure that completes the mutarotation...Ch. 20.4 - Prob. 20.10KCPCh. 20.4 - Prob. 20.11P
Ch. 20.4 - Prob. 20.12PCh. 20.4 - Prob. 20.13PCh. 20.4 - Prob. 20.1CIAPCh. 20.4 - Prob. 20.2CIAPCh. 20.4 - All cells in your body contain glycoproteins...Ch. 20.5 - Draw the structure of the and anomers that...Ch. 20.6 - Prob. 20.15PCh. 20.6 - Prob. 20.16PCh. 20.6 - Prob. 20.17KCPCh. 20.7 - Prob. 20.4CIAPCh. 20.7 - Prob. 20.5CIAPCh. 20.7 - Prob. 20.6CIAPCh. 20.7 - Prob. 20.7CIAPCh. 20.7 - Prob. 20.18PCh. 20.7 - Prob. 20.19PCh. 20.7 - Prob. 20.8CIAPCh. 20.7 - Prob. 20.9CIAPCh. 20.7 - Prob. 20.10CIAPCh. 20 - During the digestion of starch from potatoes, the...Ch. 20 - Prob. 20.21UKCCh. 20 - Consider the trisaccharide A, B, C shown in...Ch. 20 - Hydrolysis of both glycosidic bonds in the...Ch. 20 - Prob. 20.24UKCCh. 20 - Are one or more of the disaccharides maltose,...Ch. 20 - Prob. 20.26UKCCh. 20 - Prob. 20.27UKCCh. 20 - Prob. 20.28APCh. 20 - What is the family-name ending for a sugar?Ch. 20 - Prob. 20.30APCh. 20 - Classify the four carbohydrates (a)(d) by...Ch. 20 - Prob. 20.32APCh. 20 - How many chiral carbon atoms are there in each of...Ch. 20 - Prob. 20.34APCh. 20 - Prob. 20.35APCh. 20 - Name four important monosaccharides and tell where...Ch. 20 - Prob. 20.37APCh. 20 - Prob. 20.38APCh. 20 - What is the structural relationship between...Ch. 20 - Prob. 20.40APCh. 20 - In Section 15.6, you saw that aldehydes react with...Ch. 20 - Sucrose and D-glucose rotate plane-polarized light...Ch. 20 - Prob. 20.43APCh. 20 - Prob. 20.44APCh. 20 - Prob. 20.45APCh. 20 - What is mutarotation? Do all chiral molecules do...Ch. 20 - What are anomers, and how do the anomers of a...Ch. 20 - What is the structural difference between the ...Ch. 20 - D-Gulose, an aldohexose isomer of glucose, has the...Ch. 20 - Prob. 20.50APCh. 20 - In its open-chain form, D-altrose has the...Ch. 20 - Prob. 20.52APCh. 20 - Prob. 20.53APCh. 20 - Prob. 20.54APCh. 20 - Prob. 20.55APCh. 20 - What is the structural difference between a...Ch. 20 - What are glycosides, and how can they be formed?Ch. 20 - Prob. 20.58APCh. 20 - Prob. 20.59APCh. 20 - Give the names of three important disaccharides....Ch. 20 - Lactose and maltose are reducing disaccharides,...Ch. 20 - Amylose (a form of starch) and cellulose are both...Ch. 20 - Prob. 20.63APCh. 20 - Prob. 20.64APCh. 20 - Prob. 20.65APCh. 20 - Gentiobiose, a rare disaccharide found in saffron,...Ch. 20 - Prob. 20.67APCh. 20 - Prob. 20.68APCh. 20 - Prob. 20.69APCh. 20 - Amylopectin (a form of starch) and glycogen are...Ch. 20 - What is the physiological purpose of starch in a...Ch. 20 - Prob. 20.72APCh. 20 - Prob. 20.73APCh. 20 - Prob. 20.74CPCh. 20 - Prob. 20.75CPCh. 20 - Prob. 20.76CPCh. 20 - Prob. 20.77CPCh. 20 - Prob. 20.78CPCh. 20 - Write the open-chain structure of the only...Ch. 20 - Prob. 20.80CPCh. 20 - Prob. 20.81CPCh. 20 - When a person cannot digest galactose, its reduced...Ch. 20 - Describe the differences between mono-, di-, and...Ch. 20 - Prob. 20.84CPCh. 20 - Prob. 20.85CPCh. 20 - Many people who are lactose intolerant can eat...Ch. 20 - Prob. 20.87GPCh. 20 - Prob. 20.88GPCh. 20 - Prob. 20.89GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Penicillin G is a natural antibiotic that is useful for treating infections caused by Gram positive bacteria. What is the functional benefit of the semi-synthetic antimicrobials carbenicillin and ampicillin, generated by chemical modification of the R group so their R groups each are a bit different than the R group seen with penicillin G?arrow_forwardWould a homopolymer of alanine be more likely to form an α helix in water or in a hydrophobic medium? Explain.arrow_forwardSome bacteria produce poly-γ-glutamic acid, a polymer in which the amino group of each glutamate residue is condensed with the γ-carboxylate group of the adjacent residue. Draw the repeating structure of this polymer.arrow_forward
- Talose is the C4 epimer of mannose.draw the chair conformation of D-talopyranosearrow_forwardWhen cytosine is treated with bisulfite, the amino group is replaced with a carbonyl group. Identify the resulting base.arrow_forwardD-Talose is a C2 epimer of D-galactose. Using the Fischer projection structure, draw the product of reaction ofD-talose with the given reagent or enzyme and write the systematic name of the product. 1. 2,4-DNP 2. Reaction with aminotransferase and then acetyltransferase at C4arrow_forward
- You have isolated from a rare fungus an octapeptide that prevents baldness and you wish to determine its amino acid sequence. The amino acid composition is K2, D, Y, F, G, S, A. Reaction of the intact peptide with dansyl chloride yields dansyl-A. Cleavage with trypsin yields peptides whose compositions are: (K, A, S) and (G, F, K) plus a dipeptide. Reaction with chymotrypsin releases free D, a tetrapeptide with composition (k, S, F, A) and a tripeptide whose composition following acid hydrolysis is (G, K, Y). The enzymatic digests are each carried out on the whole, undansylated peptide.What is the sequence?arrow_forwardDraw the structure of a D-glucose molecule linked to threonine via a β-glycosidic linkage.arrow_forwardSelect the term in column B which best matches the description in column A. There are more terms then needed. Column A Column B 1. The pentose sugar found in DNA. a) Cis fatty acid b) Trans fatty acid c) Coenzyme d) Cofacipr 2. Commonly referred to as "insoluble fiber". 3. Considered to be an amphipathic molecule. 4. A molecule with the formula C18H3602 is probably a e) Phospholipid 5. Bond created during the formation of the primary structure of a protein. ) Glycogen 6. A non-protein organic molecule needed for proper enzyme functioning. g) Cellulose 7. Pyrimidine base found only in RNA. h) Galactose i) Fructose 8. Increasing the number of these molecules in the cell membrane would increase the permeability of the cell membrane. i) Thymine k) Uracil 9. Bond which connects nucleotide monomers together. 1) Fatty acid m) Disaccharide n) Peptide bond o) A-Helix p) B-Pleated Sheet q) Ribose r) Deoxyribose 10.arrow_forward
- Why is a strong reducing agent, such as NaBH4, not suitable for use in the conjugation of an oligosaccharide to a protein?arrow_forwardA schematic diagram of the helical structure of cytochrome b562 is reproduced below. Thisprotein belongs to the family of -proteins that have a four-helix bundle. Number the helices 1 – 4according to their N C direction. Indicate relative orientations of the macrodipoles of helices 1 – 4adjacent to the diagram of cytochrome b562. Remember that according to the definition of a dipolethat the arrow points towards the positive end.arrow_forwardWhat type of glycosidic bond is shown below, α or β?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Metabolic Pathways; Author: Wisc-Online;https://www.youtube.com/watch?v=m61bQYio9ys;License: Standard Youtube License