
a)
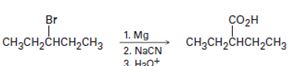
Interpretation:
The flaw in the synthetic scheme given is to be identified.
Concept introduction:
To identify:
The flaw in the synthetic scheme given.
b)
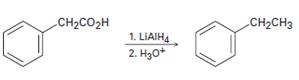
Interpretation:
The flaw in the synthetic scheme given is to be identified.
Concept introduction:
Carboxylic acids are reduced to alcohols and not to
To identify:
The flaw in the synthetic scheme given.
c)
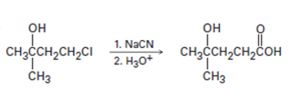
Interpretation:
The flaw in the synthetic scheme given is to be identified.
Concept introduction:
Alkyl halides when treated with cyanides yield nitriles which upon hydrolysis give carboxylic acids. Tertiary alcohols get dehydrate to yield
To identify:
The flaw in the synthetic scheme given.
Trending nowThis is a popular solution!

Chapter 20 Solutions
Organic Chemistry
- Show the difference between SN1 and E1 Reactions ?arrow_forwardShow how HC≡CH, CH3CH2Br, and (CH3)2CHCH2CH2Br can be used to prepare CH3CH2C≡CCH2CH2CH(CH3)2. Show all reagents, and use curved arrows to show movement of electron pairs.arrow_forwardList the numbers for the appropriate reagents in the correct order to accomplish the synthetic transformation shown.arrow_forward
- I need help with this organic chemistry reaction. I can use clemmensen reaction I think. If not the other options I can use Raney-nickel reduction or catalytic hydrogenation or coupling reactions or heck or Suzuki reactions can i also have an explanation. I am feeling a bit lostarrow_forwardFill in the missing reagents in the boxes below. some steps may require more than one step.arrow_forwardSN1 and E1 reactions commonly compete and form a mixture of both types of products. True or False 2. The most substituted alkene or the "Zaitsev" product is formed for the E2 elimination unless the Beta H and the LG do not have an anti relationship to each other. True or Falsearrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

