
(a)
Interpretation:
The synthesis of carbinoxamine has to be shown.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
Grignard reagent is reaction with carbonyl compound such as
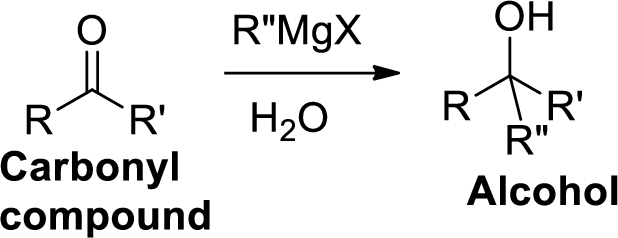
(b)
Interpretation:
The chirality of Carbinoxamine is to be identified and the possible stereoisomers has to be identified in the given synthesis.
Concept introduction:
Chiral:
A molecule is non superimposable on its mirror image is called chiral molecule.
Four different atoms attached to a carbon atom is called chiral molecule.
Isomer:
A molecule having the same molecular formula but with different chemical structure is called isomer.
Stereoisomers:
Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
Enantiomers:
A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers:
A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture:
A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
Organic Chemistry
- Following is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardMethamphetamine is a long-lasting, potent stimulant sold as a street drug. The synthesis is quite simple and one step in the synthesis is shown below. Using your knowledge of the reactions of amines, provide all the reagents necessary to complete the reaction below. Provide the reagents for step 1 and step 2 ,arrow_forwardBenzene underwent a Friedel–Crafts acylation followed by a Wolff–Kishner reduction. The product gave the following 1H NMR spectrum. What acylchloride was used in the Friedel–Crafts acylation?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
