
(a)
Interpretation:
The reagent has to be proposed for the conversion of A to B.
Concept introduction:
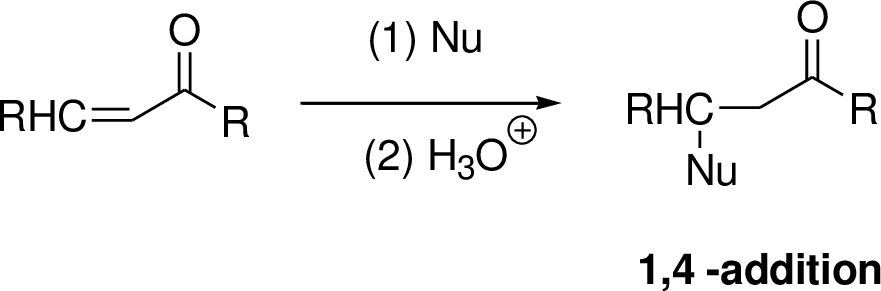
Michael reaction:
The nucleophile is reaction with α,β-unsaturated carbonyl compound (1,4 –addtion) which yield the addition product is called Michael reaction.
(b)
Interpretation:
The reagent has to be proposed for the conversion of B to C.
Concept introduction:
Reduction:
(c)
Interpretation:
The reagent has to be proposed for the conversion of C to D.
Concept introduction:
Chlorination: Alcohols reaction with thionyl chloride gives chlorinated compound.
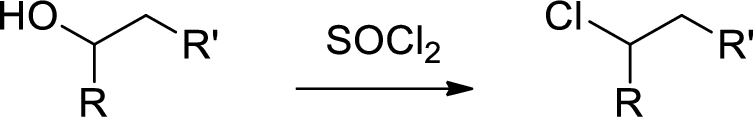
(d)
Interpretation:
The mechanism has to be proposed for the conversion of E to F and the formation of methyl chloride has to be shown through mechanism.
(e)
Interpretation:
The reagent has to be proposed for the conversion of F to Fluoxetine.
Concept introduction:
Hydrolysis: Amide undergoes hydrolysis using base like sodium hydroxide gives acid.
(f)
Interpretation:
The possible stereoisomer’s has to be shown if the product is chiral.
Concept introduction:
Chiral:
A molecule is non superimposable on its mirror image is called chiral molecule.
Four different atoms attached to a carbon atom is called chiral molecule.
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Stereoisomers: Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
Organic Chemistry
- Suggest a suitable mechanism for this multi-step reaction, and give products A and Barrow_forwardPropose a synthesis pathway for the production of the following compounds (d) to (e) from the given starting materials plus necessary organic and inorganic reagents.arrow_forwardProvide a mechanism for the following transformationarrow_forward
- In the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces sleep when it is administered to animals in large doses, paraldehyde is used as a sedative or hypnotic. Propose a mechanism for the formation of paraldehyde.arrow_forwardMethamphetamine is a long-lasting, potent stimulant sold as a street drug. The synthesis is quite simple and one step in the synthesis is shown below. Using your knowledge of the reactions of amines, provide all the reagents necessary to complete the reaction below. Provide the reagents for step 1 and step 2 ,arrow_forwardTranexamic acid, a drug useful against blood clotting, is prepared commercially from p-methylbenzonitrile. Following is one of the steps in its synthesis, draw the structure of the product of this step.arrow_forward
- propose a mechanism for the following transformationsarrow_forwardPropose a synthesis for the following transformationarrow_forwardThe ketone shown was prepared in a three-step sequence from ethyl trifluoroacetate. The first step in the sequence involved treating ethyl trifluoroacetate with ammonia to give compound A. Compound A was in turn converted to the desired ketone by way of compound B. Fill in the missing reagents in the sequence shown, and give the structures of compounds A and B.arrow_forward
- Propose a mechanism for the reaction of benzyl acetate with methylamine. Label theattacking nucleophile and the leaving group, and draw the transition state in which theleaving group leaves.arrow_forwardDevise a concise synthesis for the following transformations. Clearly show the reagent and productfor each step. Each synthesis can be accomplished in two steps except for d, which requires three steps.arrow_forwardExplain in detail which of the following compounds (A or B) when reacted with acetophenone under basic conditions will undergo a 1,4 addition reaction. Predict the product of the reaction and propose a mechanism.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
