
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.12, Problem 2.22P
Choose the more basic member of each pair of isomers, and show why the base you chose is more basic.
a. 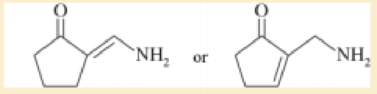
SOLUTION
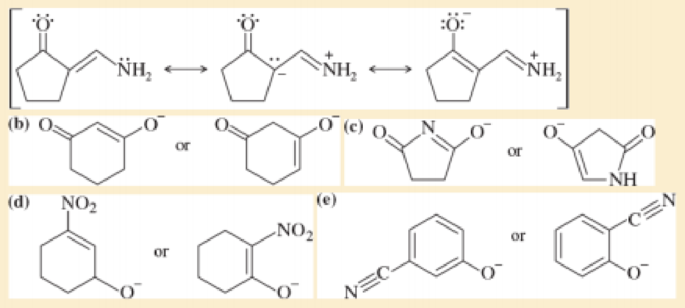
to (a) The first (left) compound is less basic because it is resonance-stabilized (like an amide), and protonation of its NH2 group would destroy that resonance Moreover, the lone pair on the NH: group is shared with the carbonyl group and the —NH2 group carries a partial positive charge The compound on the right is an
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the following compounds in order of increasing acidity (1 = least acidic, 3 = most acidic) and in the space provided use resonance (of the conjugate base) to explain why the compound you have labelled “3” is the most acidic.
2. Answer ALL parts
a)
i.
11.
NH₂
C
NH₂
NH₂
Br
E
n
NH₂
F
Rank the compounds shown above in order of INCREASING basicity
(least to most basic).
Account for the differences in basicity.
4.
Arrange the compounds in the following set in order of increasing basicity (1-the most basic; 3-the
least basic). Explain your answer!
Magnesium hydroxide, magnesium acetate, methylmagnesium bromide
Chapter 2 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 2.1A - Prob. 2.1PCh. 2.1B - The NF bond is more polar than the NH bond: but...Ch. 2.1B - For each of the following compounds 1. Draw the...Ch. 2.1B - Two isomers of 1,2-dichloroethene are known One...Ch. 2.2C - Prob. 2.5PCh. 2.2C - Prob. 2.6PCh. 2.3 - Prob. 2.7PCh. 2.4 - Calculate the pH of the following solutions a....Ch. 2.6A - Ammonia appears in Table 2-2 as both an acid and a...Ch. 2.7 - Write equations for the following acid-base...
Ch. 2.7 - Ethanol, methylamine. and acetic acid are all...Ch. 2.8 - Prob. 2.12PCh. 2.10 - Write equations for the following acid-base...Ch. 2.10 - Rank the following acids in decreasing order of...Ch. 2.11 - Prob. 2.15PCh. 2.11 - Prob. 2.16PCh. 2.11 - Consider each pair of bases and explain which one...Ch. 2.12 - Which is a stronger base ethoxide ion or acetate...Ch. 2.12 - Prob. 2.19PCh. 2.12 - Prob. 2.20PCh. 2.12 - Prob. 2.21PCh. 2.12 - Choose the more basic member of each pair of...Ch. 2.14 - Prob. 2.23PCh. 2.15D - Classify the following hydrocarbons and draw a...Ch. 2.16D - Prob. 2.25PCh. 2.17C - Draw a Lewis structure and classify each of the...Ch. 2.17C - Circle the functional groups in the following...Ch. 2 - The CN triple bond in acetonitrile has a dipole...Ch. 2 - Prob. 2.29SPCh. 2 - Sulfur dioxide has a dipole moment of 1.60 D....Ch. 2 - Which of the following pure compounds can form...Ch. 2 - Predict which member of each pair is more soluble...Ch. 2 - Prob. 2.33SPCh. 2 - Prob. 2.34SPCh. 2 - Predict which compound in each pair has the higher...Ch. 2 - All of the following compounds can react as acids...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - The Ka of phenylacetic acid is 5 2 105, and the...Ch. 2 - The following compound can become protonated on...Ch. 2 - The following compounds are listed in increasing...Ch. 2 - Prob. 2.42SPCh. 2 - Prob. 2.43SPCh. 2 - Compare the relative acidity of 1-molar aqueous...Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - The following compounds can all react as bases. a....Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - Prob. 2.48SPCh. 2 - Methyllithium (CH3Li) is often used as a base in...Ch. 2 - Label the reactants in these acid-base reactions...Ch. 2 - In each reaction, label the reactants as Lewis...Ch. 2 - Prob. 2.52SPCh. 2 - Each of these compounds can react as a nucleophile...Ch. 2 - Prob. 2.54SPCh. 2 - Give a definition and an example for each class of...Ch. 2 - Circle the functional groups in the following...Ch. 2 - Prob. 2.57SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Place the following compounds in order of relative acidity. Consider 1 to be the most acidic and 6 to be the least acidic.arrow_forwardXVI. Which of the two nitrogens (a or b) would be more basic (stronger base)? CONCISELY explain why. H ·N· of aarrow_forwardPut the following compounds in order from weakest acid to strongest acid. Explain (pKa values are not enough and trends should be considered).arrow_forward
- 5. Tropone (A) is much stronger base (conjugate acid more stabilized) than the open chain, conjugated ketone (B), both shown below. Please explain briefly why this observation makes sense. H2C CCH2 A Вarrow_forwardAccording to the following list, order the compounds in increasing order with respect to their acidity. For each compound, indicate in the structure which is the acidic hydrogen. Please solve correctly.arrow_forwardb) Which of the following compounds would you expect to be most acidic? Explain your answer, including clear drawing/analysis of the relevant conjugate base(s). H.arrow_forward
- For compound A below, draw at least 6 different constitutional isomers. (Hint you don't have to count the hydrogens on every structure if you have the right number of non-hydrogen atoms and degrees of unsaturation.) OH OH Use the constitutional isomers of A you drew above or others, to find a constitutional isomer that matches each description below. Use your understanding of Acid-Base principles to demonstrate the difference in pKa between each isomer and A. (Hint: what can change when you draw a different constitutional isomer that affects the pKa?) a. An isomer that is at least 105 times less acidic than A b. An isomer that is comparable in acidity to A c. An Isomer that is at least 105 times more acidic than Aarrow_forwardB). Pyridine and pyrrole (shown below) are considered to be aromatic compounds. i. Explain why this is so using orbital diagrams to illustrate your answer. and N-H ii Explain which of the two is a stronger base and explain why.arrow_forwarda) Draw the conjugate base of the compound shown. Include all resonance structures. b) The pKa of phenol is 10. Does the compound shown have a pKa greater or less than 10? Fully explain your answer. (Hint: “It’s more acidic” or “it’s less acidic” is not a full explanation)arrow_forward
- Which compound is more acidic and why? I thought it would be the right, because nitrogen has a hydrogen attached to it, but oxygen is more electronegative than nitrogen, but doesn't have a hydrogen readily available (other than the methyl group on the other side).arrow_forwardRank the following in order of acidity with 1 being the most acidicarrow_forwardWhich is a stronger acid and has the most stable conjugate base? Estimate the pKA values for both compounds.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY