
Concept explainers
Rank the compounds in each group in order of increasing frequency of the
a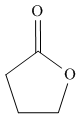
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
Organic Chemistry (Looseleaf) - With Access
- Assume a compound with the formula C4H8O. a) How many double bonds and/or rings does your compound contain? b) If your compound shows an infrared absorption peak at 1715 cm-1, what functional group does it have? c) If your compound shows a single 1H NMR absorption peak at 2.1 δ, what is its structure?arrow_forwardThe UV spectrum for which of the following compounds would have the largest value for λ max? A. (CH3)3CCHO B. CH3CH2CH2CH2OH C. CH3CH2OCH2Ph D. CH3CH2CH2COOH E. PhCH2C(O)CH3arrow_forwardWhich group in each pair is assigned the higher priority? a. – CH3, – CH2CH3 b. – I, – Br c. – H, – D d. – CH2Br, – CH2CH2Br e. – CH2CH2Cl, – CH2CH(CH3)2 f. – CH2OH, – CHOarrow_forward
- Rank the following compounds in order of increasing frequency of their carbonyl absorption in the infrared.arrow_forwardTwo products, A and B, are obtained from the reaction of 1-bromobutane with NH3. Compound A reacts with acetyl chloride to form C, and compound B reacts with acetyl chloride to form D. The IR spectra of C and D are shown. Identify A, B, C, and D.arrow_forward15. Compound C has a molecular weight of 94.54 and the 1H-NMR spectrum shows four signals - a triplet at 3.81 ppm, a triplet at 3.63 ppm, a singlet at 2.19 ppm, and a triplet of triplets at 2.02 ppm. The mass, IR, and 13C-NMR spectra of compound C are shown below, they are also downloadable for closer inspection by clicking the link under the spectral data. Identify C and explain your reasoning. refer to picturearrow_forward
- The 1H and 13C NMR spectra of compound A, C8H9Br are shown below. Answer the following questions. 1(a) Degree of the unsaturation of this compound is = , 1(b) The derived unsaturation number indicates that compound has ............= 1(c) Two peaks in between 6.5 - 8.0 δ indicate that compound is= 1(d) According to the splitting pattern of the peak at 1.20 δ and 2.58 δ indicates that compound has a .................. group= 1(e) According to the 1H NMR spectrum the number of nonequivalent aromatic proton sets in the compound = 1(f) According to the 13C NMR, the number of nonequivalent carbons in the compound is = 1(g) According to your answer in Q 1(f) the compound has a plane of symmetry Yes or NO = 1(h) The IUAC name for this unknown compound isNOT TOO SURE ABOUT MY ANSWERS, PLEASE CORRECT ME IF I'M WRONGarrow_forwardWhich of the following molecules would have 5 peaks in its 13C NMR spectrum? (Could be multiple) a.) cyclohexane b.) 3-methylpentane c.) 1,3-dimethylcyclobutane d.) methylcyclohexane e.) 2,2-dimethylpentane f.) 1-chloro-2-methylcyclopentanearrow_forward1) A compound with molecular formula C49H96 could not be a ... 3)Compound Y has molecular formula C4H8O and its IR spectrum shows no absorption above 3000 cm-1 and no absorption between 1680 and 1840cm-1. Which of the following could be compound Y? 4) Which of the following will have its carbonyl absorption in its IR spectrum at the lowest wavelength? Please explain 1,3,4 thank youarrow_forward
- Treatment of 3,4-dibromohexane with strong base leads to loss of 2 equivalents of HBr and formation of a product with formula C6H10. Three products are possible. Name each of the three, and tell how you would use 1H and 13CNMR spectroscopy to help identify them. How would you use UV spectroscopy?arrow_forwardAssign as many peaks as possible in the unknown #1 (C8H8O) IR Spectrum.arrow_forwardPropose a structure consistent with each set of data Compound B:Molecular formula: C9H10O2 IR absorption at 1669 cm−1 1H NMR data: 2.5 (singlet, 3 H), 3.8 (singlet, 3 H), 6.9 (doublet, 2 H),and 7.9 (doublet, 2 H) ppmarrow_forward
