
Interpretation:
The products that would be formed on salicin reacting with dilute aqueous HCl is to be determined and the mechanism of reaction for the this reaction is to be outlined.
Concept introduction:
舧 Electrophiles are electron-deficient species, which has positive or partially positive charge. Lewis acids are electrophiles, which accept electron pair.
舧 Nucleophiles are electron-rich species, which has negative or partially negative charge. Lewis bases are nucleophiles, which donate electron pair.
舧 Substitution reaction: A reaction in which one of the hydrogen atoms of a hydrocarbon or a
舧 Elimination reaction: A reaction in which two substituent groups are detached and a double bond is formed is called elimination reaction.
舧 Addition reaction: It is the reaction in which unsaturated bonds are converted to saturated molecules by the addition of molecules.
舧 A carbohydrate is a
舧 Salicin is a naturally occurring glycoside and is originally found in the bark of willow trees. It is an effective analgesic for relieving pain. Its structure is given below:
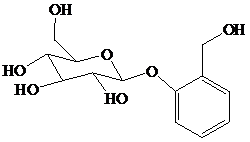
舧 Glycosides are carbohydrate acetals and can be simple or complex. These molecules are stable in basic solutions, but produce sugar and alcohol in acidic solutions.
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
EBK ORGANIC CHEMISTRY
- Predict the products obtained from the reaction of triolein with the following reagents.(a) NaOH in water (b) H2 and a nickel catalyst (c) Br2 in CCl4arrow_forward(b) Suggest a reasonable biosynthesis for the naturally occurring alkaloid coniine (isolated from hemlock), starting from octanoic acid. Coniinearrow_forward(D)The best route to synthesise aromatic primary amines is by reduction of the corresponding nitro compounds. Draw the reaction scheme for the preparation of p-toluidine.arrow_forward
- Synthesize octadecyl stearate using linoleic acid (18:2) (Given, stearic acid: 18:0) as the only starting material.arrow_forwardShow how to synthesize the following amines from the indicated starting materials.(a) N-cyclopentylaniline from aniline (b) N-ethylpyrrolidine from pyrrolidinearrow_forwardBased on the image attached, it shows methyl salicylate reacts with Ethanamine, and Ether act as a solvent to form N-ethylbenzamide as a product. Explains the mechanism reaction of conversion ester to an amide.arrow_forward
- Predict the products formed when limonene reacts with the followingreagents.(a) excess HBr (b) excess HBr, peroxides (c) excess Br2 in CCl4(d) ozone, followed by dimethyl sulfidearrow_forwardGive one chemical test to distinguish between the following pairs of compounds : (i) Methylamine and dimethylamine (ii) Secondary and tertiary amines (iii) Ethylamine and aniline (iv) Aniline and benzylamine (v) Methylamine and methanol (vi) Methylamine and N, N-Dimethylamine (vii) Ethanol and ethanaminearrow_forward22.47 Tertiary amines with three different alkyl groups are chiral but cannot be resolved because pyramidal inversion causes racemization at room temperature. Nevertheless, chiral aziridines can be resolved and stored at room temperature. Aziridine is a three-membered heterocycle containing a nitrogen atom. The following is an example of a chiral aziridine. In this compound, the nitrogen atom is a chiral center. Suggest a reason why chiral aziridines do not undergo racemization at room temperature.arrow_forward
- Predict the products from the reactions of the following amines with sodium nitrite indilute HCl.(a) cyclohexanaminearrow_forwardWrite a structural formula for each of the following compounds: (a) m-Chlorobenzoyl chloride (b) Trifluoroacetic anhydride (c) cis-1,2-Cyclopropanedicarboxylic anhydride (d) Ethyl cycloheptanecarboxylate (e) 1-Phenylethyl acetate (f) 2-Phenylethyl acetate (g) p-Ethylbenzamide (h) N-Ethylbenzamide (i) 2-Methylhexanenitrilearrow_forward(a) Explain why phentermine [PhCH2C(CH3)2NH2] can’t be made by a reductive amination reaction.(b) Give a systematic name for phentermine, one of the components of the banned diet drug fen–phen.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
