
ORGANIC CHEMISTRY (LL)-W/ACCESS
10th Edition
ISBN: 9781259717536
Author: Carey
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.22, Problem 17P
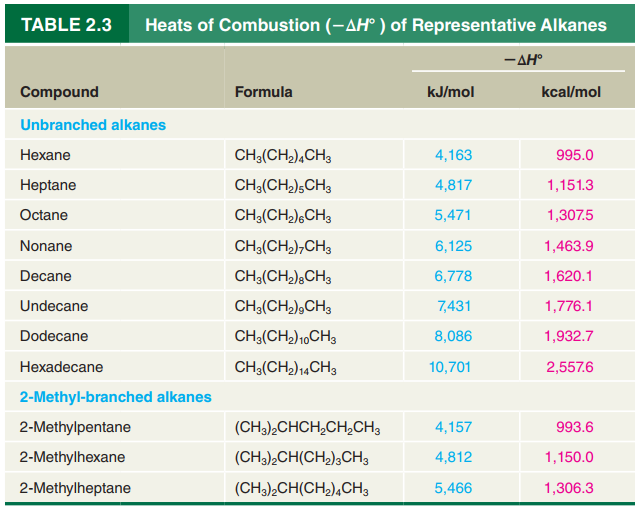
Using the data in Table
Icosane (in kJ/mol)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
If a butane reaction only generated 135kJ, how many grams of butane was consumed?
Need help solving this problem.
calculating heat of combustion of quinone
The heat of combustion for C2H6 (30.07 g/mol) is -1559.5 kJ/mol. Calculate the heat of reaction (in kJ) when 8.24 g C2H6 is combusted. Be sure to include the correct sign in your answer (+ or -) and do not put a space between the sign and the number. Do not include units in your answer. Round answer to the hundredths place.
Chapter 2 Solutions
ORGANIC CHEMISTRY (LL)-W/ACCESS
Ch. 2.4 - Prob. 1PCh. 2.7 - Prob. 2PCh. 2.8 - Identify the orbital overlaps of all of the bonds...Ch. 2.9 - The hydrocarbon shown, called vinylacetylene, is...Ch. 2.12 - Prob. 5PCh. 2.12 - Prob. 6PCh. 2.13 - Prob. 7PCh. 2.14 - Refer to Table 2.2 as needed to answer the...Ch. 2.15 - Prob. 9PCh. 2.15 - Prob. 10P
Ch. 2.16 - Prob. 11PCh. 2.17 - Prob. 12PCh. 2.18 - Prob. 13PCh. 2.20 - Prob. 14PCh. 2.21 - Match the boiling points with the appropriate...Ch. 2.22 - Write a balanced chemical equation for the...Ch. 2.22 - Using the data in Table 2.3, estimate the heat of...Ch. 2.22 - Prob. 18PCh. 2.22 - Prob. 19PCh. 2.23 - Prob. 20PCh. 2.23 - Which of the following reactions requires an...Ch. 2 - The general molecular formula for alkanes is...Ch. 2 - Prob. 23PCh. 2 - Prob. 24PCh. 2 - Prob. 25PCh. 2 - What is the hybridization of each carbon in...Ch. 2 - Prob. 27PCh. 2 - Does the overlap of two p orbitals in the fashion...Ch. 2 - Prob. 29PCh. 2 - Aphids secrete an alarm pheromone having the...Ch. 2 - All the parts of this problem refer to the alkane...Ch. 2 - Prob. 32PCh. 2 - Prob. 33PCh. 2 - Prob. 34PCh. 2 - From among the 18 constitutional isomers of C8H18,...Ch. 2 - Give the IUPAC name for each of the following...Ch. 2 - Using the method outlined in Section 2.16, give an...Ch. 2 - Prob. 38PCh. 2 - Write a balanced chemical equation for the...Ch. 2 - The heats of combustion of methane and butane are...Ch. 2 - In each of the following groups of compounds,...Ch. 2 - Given H for the reaction H2(g)+12O2(g)H2O(l)...Ch. 2 - Prob. 43PCh. 2 - Prob. 44PCh. 2 - Prob. 45PCh. 2 - Prob. 46PCh. 2 - Prob. 47PCh. 2 - Compound A undergoes the following reactions:...Ch. 2 - Prob. 49PCh. 2 - Some Biochemical Reactions of Alkanes Alkanes...Ch. 2 - Prob. 51DSPCh. 2 - Some Biochemical Reactions of Alkanes Alkanes...Ch. 2 - Prob. 53DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using the heats of formation below, calculate the heat of reaction for the following reaction: C2H6(g) + 7/2O2(g) → 2CO2(g) + 3H2O(ℓ) ΔHf° (kJ/mol) C2H6(g): -84.86 O2(g): 0 CO2(g): -393.5 H2O(ℓ): -285.8arrow_forwardUsing the following equation for conversion of octane, calculate the heat associated with the combustion of 150.0g of the octane assuming complete combustion. The molar mass of octane is 114.33 g/mol. The molar mass of oxygen is 31.9988 g/ mole 2 C8H18+25O2 —> 16 CO2+ 18 H2O H degree rxn = -11018arrow_forwardDraw a enthalpy diagram on the combustion of a butane gas canisterarrow_forward
- Combustion reactions are exothermic. The heat of reaction for the combustion of 2-methylhexane, C7H16, is 1.150×103 kcal/mol. What is the heat of combustion for 2-methylhexane in kcal/gram? kcal/gramHow much heat will be given off if molar quantities of 2-methylhexane react according to the following equation? C7H16 + 11 O27 CO2 + 8 H2O kcalarrow_forwardCombustion: C2H6(g)+O2(g)--->(heat)_____________ do not include heat in your answerarrow_forwardWhat would be the heat of combustion of C4H10(g) if the heats of formation of C4H10(g), CO2(g), and H2O(l) are -124.73 kJ/mol, -393.5 kJ/mol, and -285.8 kJ/mol, respectively.C4H10(g) + 13/2O2(g) ↔ 4CO2(g) + 5H2O(l)arrow_forward
- The heat of combustion for C2H4 (28.05 g/mol) is -1411 kJ/mol. Calculate how much heat is released (in kJ) when 11.66 g C2H4 is combusted. Be sure to report your answer as a positive value and do not put a space between the sign and the number. Do not include units in your answer. Report your answer to the hundredths placearrow_forwardGUIDE QUESTIONS: Calculate the heat of combustion of the following reactions (show the computation). Use the enthalpy of formation of the substances given below: Substances CO2 (g) -393.5 H2O (l) -285.8 O2 (g) 0 CH3CH2OH -276.98 CH3(CH2)2OH -303.3 CH3(CH2)3OH -332.8 a. combustion of ethanol b. combustion of propanol c. combustion of butanolarrow_forwardMethanol undergoes combustion according to the equation. Find the amount of heat given off from the combustion of 64 g CH3OH (methanol). (MM = 32 g/mol) (∆H=-1452.8 kJ) 2 CH3OH(l) + 3 02(g) -> 2 C02(g) + 4 H2O(l) A. - 726.4 kJ B. -1452.8 kJ C. -2179.2 kJ D. -2905.6 kJarrow_forward
- Methanol undergoes combustion according to the equation. Find the amount of heat given off from the combustion of 64 g CH3OH (methanol). (MM = 32 g/mol) (∆H=-1452.8 kJ) 2 CH3OH(l) + 3 O2(g) -> 2 CO2(g) + 4 H2O(l) a. 726.4 kJ b. -1452.8 kJ c. -2179.2 kJ c. -2905.6 kJarrow_forwardFind the total amount of heat required to heat 15.00 mL of acetone (d=0.7845 g/mL M.W=58.080 g/mole m.p=- 94.70 deg *C; b.p.=56.05*C)from 25.00*C to 60 deg *Carrow_forwardHow much heat is produced by the complete reaction of 6.81 kg of nitromethane? Nitromethane (CH3NO2) burns in air to produce significant amounts of heat via the following reaction: 2CH3NO2(l)+32O2(g)→2CO2(g)+3H2O(l)+N2(g)ΔH∘rxn=−1418kJarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY