
(a)
Interpretation: The major products are to be found when the given compounds are treated with excess methyl iodide followed by aqueous silver oxide and heat.
Concept Introduction:
The reagents such as methyl iodide and silver oxide are used as the reaction conditions which are called Hofmann elimination. The
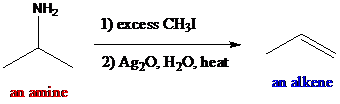
To find: The major product of cyclohexyl amine in Hofmann elimination
Draw and analyze the structure of cyclohexyl amine
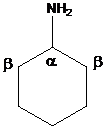
(b)
Interpretation: The major products are to be found when the given compounds are treated with excess methyl iodide followed by aqueous silver oxide and heat.
Concept Introduction:
The reagents such as methyl iodide and silver oxide are used as the reaction conditions which are called Hofmann elimination. The amines undergo exhaustive alkylation in the presence of excess methyl iodide producing quaternaryammonium salt which is excellent leaving group. After treatment with strong base, aqueous silver oxide and heat, the quaternary ammonium salt provides alkenes. Alkene formation involves in
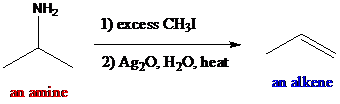
To find: The major product of (R)-3-methyl-2-butanamine in Hofmann elimination
Draw and analyze the structure of (R)-3-methyl-2-butanamine
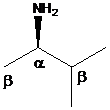
(c)
Interpretation: The major products are to be found when the given compounds are treated with excess methyl iodide followed by aqueous silver oxide and heat.
Concept Introduction:
The reagents such as methyl iodide and silver oxide are used as the reaction conditions which are called Hofmann elimination. The amines undergo exhaustive alkylation in the presence of excess methyl iodide producing quaternaryammonium salt which is excellent leaving group. After treatment with strong base, aqueous silver oxide and heat, the quaternary ammonium salt provides alkenes. Alkene formation involves in
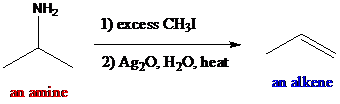
To find: The major product of N,N-dimethyl-1-phenylpropan-2-amine in Hofmann elimination
Draw and analyze the structure of N,N-dimethyl-1-phenylpropan-2-amine.
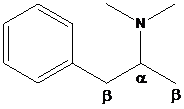
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
Organic Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





