
Concept explainers
(a)
Interpretation:
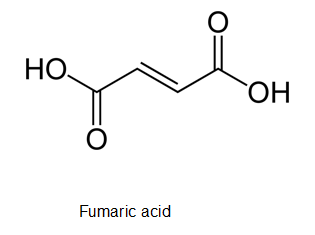
The empirical formula of fumaric acid in which the elemental composition is 41.4% C, 3.5 % H and 55.1 % O needs to be determined.
Concept introduction:
Empirical formula is the simplest formula of any organic or inorganic compound that represents the simple ratio of all atoms present in the molecule. It can be calculated with the help of elemental composition of molecule. Certain steps must be used to get the empirical formula:
- Consider mass % as mass in grams and calculate moles of element with the help of molar mass
- Calculate the moles of each element in least whole number
- Write the number of each atom as subscript to write the empirical formula.
(b)
Interpretation:
The molecular weightof fumaric acidneeds to be determined.
Concept introduction:
Osmotic pressure is a colligative property that depends on the number of solute particles in the solution. It is mainly used to calculate the molecular weight of substance dissolve in certain volume of solvent. The relation between osmotic pressure and concentration can be shown as:
(c)
Interpretation:
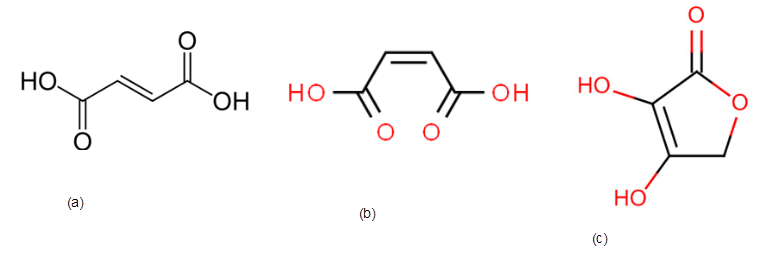
Thethree possible structures of fumaric acid if the empirical formula is CHO and molar mass is 116 g/mol needs to be determined.
Concept introduction:
Empirical formula is the simplest formula of any organic or inorganic compound that represents the simple ratio of all atoms present in the molecule. It can be calculated with the help of elemental composition of molecule. Certain steps must be used to get the empirical formula:
- Consider mass % as mass in grams and calculate moles of element with the help of molar mass
- Calculate the moles of each element in least whole number
- Write the number of each atom as subscript to write the empirical formula.
(d)
Interpretation:
The correct structure of fumaric acid if it contains a trans double bond needs to be determined.
Concept introduction:
Empirical formula is the simplest formula of any organic or inorganic compound that represents the simple ratio of all atoms present in the molecule. It can be calculated with the help of elemental composition of molecule. Certain steps must be used to get the empirical formula:
- Consider mass % as mass in grams and calculate moles of element with the help of molar mass
- Calculate the moles of each element in least whole number
- Write the number of each atom as subscript to write the empirical formula.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
LCPO CHEMISTRY W/MODIFIED MASTERING
- Please answer question number 2arrow_forwardExplain why cholesterol, a compound with molecular formula C 27H 46O and one OH group, is soluble in CCl 4 but insoluble in water.arrow_forwardPredict the water solubility of each compound: (a) KCl; (b) methanol (CH 3OH); (c) hexane (C 6H 14).arrow_forward
- Ascorbic acid has a chemical formula of C6H8O6 and is water soluble. A solution containing 80.5 g of ascorbic acid is dissolved in 210g of water. The resulting solution has a density of 1.22g/mL at 55°C. Calculate the mass percentage of the ascorbic acid in the solution.arrow_forwardWhich statements accurately describe soap? Select one or more: Soaps are a mixture of fatty acid salts and glycerol. Soaps react with ions in hard water to create a precipitate. Soaps should be weakly alkaline in solution. aps work in solutions of any pH. Soaps are both hydrophobic and hydrophilic.arrow_forwardAnswer no. 18arrow_forward
- Ethanoic acid (acetic acid) is verysoluble in water. However, naturally occurring longchaincarboxylic acids, such as palmitic acid(C H 3 (C H 2 ) 14 COOH), are insoluble in water. Explain.arrow_forwardA mixture of ethanol, benzaldehyde and KMnO4 is mixed together. Would you expect a precipitate to form based on the reactants functional groups?arrow_forwardWhat is the molarity of an ethylacetate solution that contains 2.54 moles C4H8O2 dissolved in 600.0 mL of solution?arrow_forward
- Arrange the following acids in the increasing order of their solubility in water: p-ClC6H4COOH, CH3COOH, CH3(CH2)3COOH. Explain why their order became like that?arrow_forward90 g of KCl is dissolved in 250 g of water at 35 celcius with 80% dissociation. Calculate the: • BPE FPD • Vapor Pressure of the solution • Osmotic Pressurearrow_forwardThe terms nonpolar and polar as well as hydrophilic andhydrophobic can also be used to describe molecules. Howare these terms related to lipid-soluble and non-lipid-soluble?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





