
Concept explainers
(a)
Interpretation:
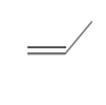
The possibility of cis-trans isomer of Prop-1-ene is to be determined and if it exists, it needs to be drawn.
Concept introduction:
The cis-trans isomers, also known as geometric isomers, are isomers which have same molecular formula but have different permanent position in space.
(b)
Interpretation:
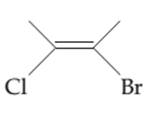
The possibility of cis-trans isomer of 2-Bromo-3-chlorobut-2-ene is to be determined and if it exists then it needs to be drawn.
Concept introduction:
The cis-trans isomers, also known as geometric isomers, are isomers which have same molecular formula but have different permanent position in space.
(c)
Interpretation:
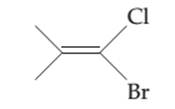
The possibility of cis-trans isomer of 1-Bromo-1-chloro-2-methylpropene is to be determined and if it exists then it needs to be drawn.
Concept introduction:
The cis-trans isomers, also known as geometric isomers, are isomers which have same molecular formula but have different permanent position in space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
LCPO CHEMISTRY W/MODIFIED MASTERING
- Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that do.arrow_forward||| O NAMING AND DRAWING ORGANIC MOLECULES Interpreting condensed chemical structures Use this condensed chemical structure to complete the table below. 0 NH₂ - CH — C — OH | CH3 The condensed chemical structure of alanine Some facts about the alanine molecule: number of carbon-carbon single (C - C) bonds: number of carbon-hydrogen single (C-H) bonds: number of oxygen-hydrogen single (O - H) bonds: number of nitrogen-hydrogen single (N - H) bonds: number of lone pairs: X Ś 0 ☐ 1 D 503 Jessarrow_forwardDraw an isomer for the molecule C4H8. Do not draw geometric isomers (i.e., cis-trans isomers).arrow_forward
- Replace one atom in CH3CH2Br by another Br atom to form a model of a compound with the formula C2H4Br2. Draw projection drawings and perspective drawings of all possible isomers. Does replacing different hydrogen atoms in CH3CH2Br to produce C2H4Br2 give different isomers? (i.e. Are all the hydrogen atoms equivalent?) If there are multiple isomers, what type of isomerism do they represent?arrow_forwardWhat kind of constitutional isomerism do the compounds have?arrow_forwardIs the compound a Cis or Trans Isomer? Name the isomer with Cis/Trans prefix.arrow_forward
- Identify which type of isomer the following structures represent. * Br Br Positional isomer Skeletal isomer Cis/trans isomer E/Z isomerarrow_forwardProjection drawings of all isomers of C6H4Cl2. Can you convert one into another without breaking any bonds? What type of isomerism do these represent?arrow_forwarda.Give all the structural isomers of the compound C3H5Cl which contains one C=C bond. (Hint: 3 isomers) b.One of the structural isomers in above question shows cis-trans isomerism. Draw and name this pair of isomers.arrow_forward
- In terms of organic chemistry how do you know if an atom has ionic bonds or covalent bonds and how do you know if it has just one of these or if it has both? Could you provide some examples of what an ionic bond looks like, covalent bond looks like, and what an atom would look like with both of these?arrow_forwardIsomers are different compounds that have the same molecular formula. If the atoms are connected in different ways, they are called constitutional or structural isomers. Geometric isomers are a type of isomer where the order of the atoms in the two compounds is the same but their arrangement in space is different. The most common types of geometric isomers are cis- and trans- isomers. PART 5: Constitutional/Structural Isomers Draw (using any method you wish) and give the IUPAC name for any four isomers with the molecular formula C6H12Brz. Structure: - Br CH₂ сна CH₂CH CH CH₂ CH₂ Br. Name: bromo-3 methyl peature Name: Structure: Structure: Name: Structure: comors Name:arrow_forwardWhich is most stable, cis-1,4-dimethylhexane, or the trans isomoer? Draw the structure of the more stable isomer (your structure should clearly distinguish between all axial and equatorial positions.)arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning




