
Concept explainers
Interpretation:
The number of electrons in the
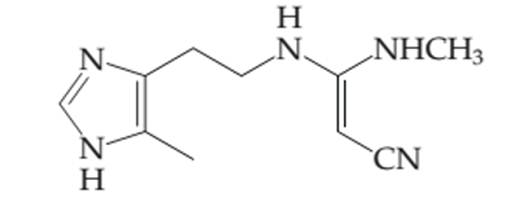
Concept introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds. The molecular formula of an organic compound represents the number of bonded atoms with their atomic symbols.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
LCPO CHEMISTRY W/MODIFIED MASTERING
- Is it possible for a motor fuel to have a negative octane rating? Explain.arrow_forwardFatty acids are carboxylic acids that have long hydrocarbon chains attached to a carboxylate group. How does a saturated fatty acid differ from an unsaturated fatty acid? How are they similar?arrow_forward12) What is the major difference between an antiaromatic and aromatic compound? A) The structure must be cyclic for aromatic but not antiaromatic compounds? B) Antiaromatic compounds have at least one sp3 hybridized atom in the ring C) Antiaromatic compounds can assume a chair-like structure while aromatic compounds are nearly flat D) Aromatic compounds cannot have a charged atom in the structure E) Only aromatic compounds follow Huckle's rule.arrow_forward
- Classify the compounds shown as aromatic, antiaromatic, or nonaromatic . Assume all the structures are planar.arrow_forwardthe bonds C-C, C-H, and C-O, in a molecule of acetone are polar covalent or nopolar covalent?arrow_forwardUsing only single bonds, how many hydrogen atoms would it take to covalently bond to one carbon atom before the molecule would become most stable?arrow_forward
- How Two molecules of acetic acid (CH3COOH) held together by twohydrogen bonds ?arrow_forwardIdentify the functional groups in the ball-and-stick model of neral, a compound with a lemony odor isolated from lemongrass ?arrow_forwardIdentify the functional groups in leukotriene C4, a major contributor to the inflammation associated with asthma.arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





