
Concept explainers
(a)
Interpretation:
The more acidic group −NH3+ or −COOH in the neutral amino acid alanine needs to be determined.
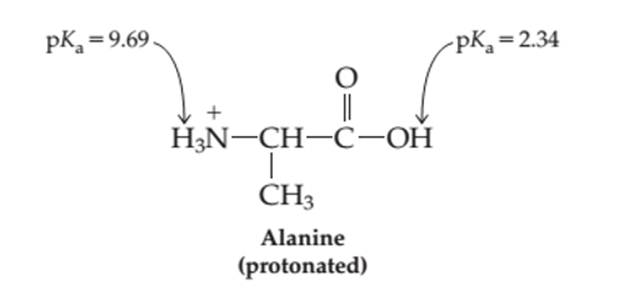
Concept introduction:
Proteins are
(b)
Interpretation:
The percentage of each form-protonates (H2A+ ), neutral (HA) and deprotonated (A- ) in the aqueous solution at pH = 4.00 needs to be determined.
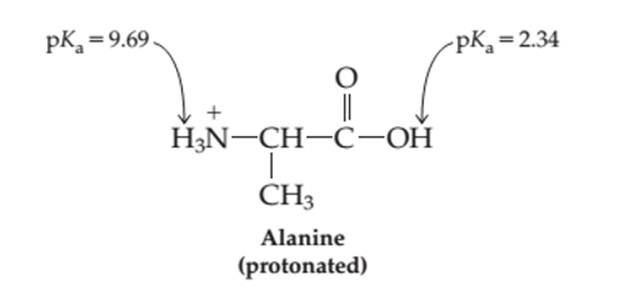
Concept introduction:
Proteins are biomolecules that are composed of certain monomer units such as amino acids. These are carboxylic acids with one amino group bonded on alpha-C atom of the molecule. These amino acids involve in condensation process to form peptides and polypeptides which further form complex protein molecules. Amino acids are the organic molecules with both
(c)
Interpretation:
The percentage of each form-protonates (H2A+ ), neutral (HA) and deprotonated (A- ) in the aqueous solution at pH = 8.50 needs to be determined.
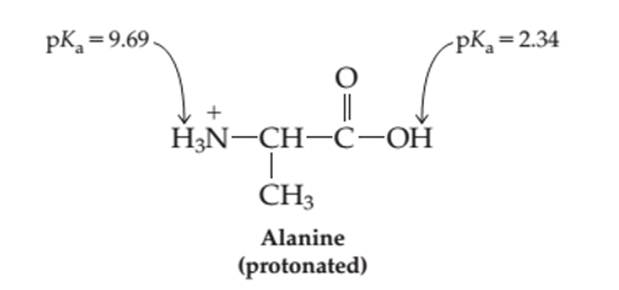
Concept introduction:
Proteins are biomolecules that are composed of certain monomer units such as amino acids. These are carboxylic acids with one amino group bonded on alpha-C atom of the molecule. These amino acids involve in condensation process to form peptides and polypeptides which further form complex protein molecules. Amino acids are the organic molecules with both
(d)
Interpretation:
The pH at which the neutral form of alanine present in the maximum amount needs to be determined.
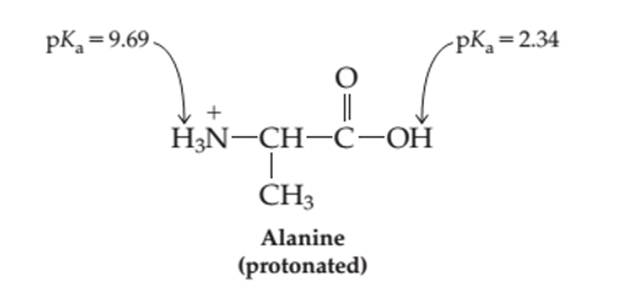
Concept introduction:
Proteins are biomolecules that are composed of certain monomer units such as amino acid. These are carboxylic acids with one amino group bonded on the alpha-C atom of the molecule. These amino acids involve in condensation process to form peptides and polypeptides which further form complex protein molecules. Amino acids are the organic molecules with both
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
CHEMISTRY-TEXT
- 16-27 Guanidine, p/Ca 13.6, is a very strong base, almost as basic as hydroxide ion. NH NH2+ II II H2N—C—NH2 + H2O H2N—C—NH2 + OH" Guanidine Guanidinium ion pKb = 13.6 Complete the Lewis structure for guanidine, showing all valence electrons. The remarkable basicity of guanidine is attributed to the fact that the positive charge on the guanidinium ion is delocalized by resonance over the three nitrogen atoms. This delocalization increases the stability of the guanidinium ion relative to the ammonium ion or substituted ammonium ions. Draw three equivalent contributing structures for the guanidinium ion and show by the use of curved arrows how these three contributors are related. Propose an explanation for the fact that protonation occurs on the C=NH nitrogen rather than on one of the —NH., nitrogens. (Hint: Consider the resonance stabilization of the structure formed by protonation on a —NH., nitrogen compared with the resonance stabilization of the structure formed by protonation on the =NH nitrogen.) Predict the N—C—N bond angles in the hybrid. Which is the stronger acid, the ammonium ion or the guanidinium ion?arrow_forwardWhat is the actual chemical component for an anthocyanin (assume a cyanidin structure), and a carotenoid (assume a lutein structure), and indicate what is the common feature of these indicators that could act as an acid or base. (While the structures are complicated molecules, compare with acetic acid, CH3COOH, a weak acid).arrow_forwardFor an acid with a pKa of 5.00, what is the pKb of its conjugate base?arrow_forward
- Write a net ionic equation to show that piperidine, C5H11N, behaves as a Bronsted-Lowry base in water.arrow_forwardThe amino acid alanine is a solid at room temperature and has a melting point of 315 °C, while pyruvic acid (CH 3COCO 2H) has a similar molecular weight but is a liquid at room temperature with a boiling point of 165 °C. Account for the difference.arrow_forwardWrite a net ionic equation to show that piperidine, C5H11N, behaves as a Bronsted-Lowry base in water. BL base BL acid BL acid BL base + H2O +arrow_forward
- Calculate the pH of a 1.01×10-2 M solution of the decongestant ephedrine hydrochloride if the pKb of ephedrine (its conjugate base) is 3.86.arrow_forwardWhere is the effective buffering range for this amino acid in the acidic region?arrow_forwardCalculate the pH of a 0.2 M solution of an amine that has a pKa of 9.5arrow_forward
- a) Find the pH of 0.1 M of the different form of histidine species. (See image attached) b) What is the principal species at pH , pH 5, pH 8, and pH 11?arrow_forwardThe monohydrogen phosphate ion HPO3-2 has a Pka of 12. One could best describe this ion as a a) very strong acid b) moderately strong acid c) moderately weak acid d) very weak acid.arrow_forwardThere are two pH values in which Glutamic acid will have an a complete net charge of 2.17? what is the lowest PH1 and highest of pH2 round your amswers to the nearest 0.1 no orher information was providedarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

