
Concept explainers
(a)
Interpretation:
The line drawing nitrogen containing compound is to be drawn and formal charge on each atom is to be shown.
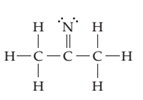
Concept introduction:
The line drawing is a chemical structure in which carbon and hydrogen molecules are not drawn. In these types of structures, lines are used to draw structure representing molecules. In the electron dot structure, the lone pair of electrons is represented as two dots on the respective symbol of the atom.
Formal charge is the charge present on each atom in the molecule; it is calculated by the following formula:
Formal Charge = Number of valence electrons on
(b)
Interpretation:
The line drawing containing compound is to drawn and formal charge on each atom is to be shown.
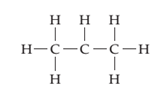
Concept introduction:
The line drawing is a chemical structure in which carbon and hydrogen molecules are not drawn. In these types of structures, lines are used to draw structure representing molecules. In the electron dot structure, the lone pair of electrons is represented as two dots on the respective symbol of the atom.
Formal charge is the charge present on each atom in the molecule; it is calculated by the following formula:
Formal Charge = Number of valence electrons on atom − [Number of non-bonded electrons + Number of bonds].
(c)
Interpretation:
The line drawing nitrogen containing compound is to drawn and formal charge on each atom is to be shown.
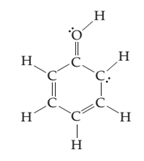
Concept introduction:
The line drawing is a chemical structure in which carbon and hydrogen molecules are not drawn. In these types of structures, lines are used to draw structure representing molecules. In the electron dot structure, the lone pair of electrons is represented as two dots on the respective symbol of the atom.
Formal charge is the charge present on each atom in the molecule; it is calculated by the following formula:
Formal Charge = Number of valence electrons on atom − [Number of non-bonded electrons + Number of bonds].
Trending nowThis is a popular solution!

Chapter 23 Solutions
CHEMISTRY-TEXT
- the formal charges on all the atoms in the following Lewis diagrams. Which one would best represent bonding in the molecule Cl2O ?arrow_forwardA complete Lewis structure must show all nonzero formal charges. Complete each of thefollowing Lewis structures by adding any missing formal charges.arrow_forwardThe oxygen atom in acetone possesses ____ unshared pairs and ____ shared pairs of electrons. The number of electrons that belong to oxygen is ____. Oxygen is a Group ____ element. The formal charge on oxygen in acetone is ____.arrow_forward
- What is the formal charge on nitrogen in the following structure?arrow_forwardProvide an arrow-pushing mechanism to explain the formation of benzoic acid from bromobenzene. (Use line drawings with lone pairs and formal charges)arrow_forwardwhat is the generic formula (format for example, AX3E) and formal charge of Chlorine trifluoride?arrow_forward
- Unshared, or lone, electron pairs play an important role in determining the chemical and physical properties of organic compounds. Thus, it is important to know which atoms carry unshared pairs.Use the structural formulas below to determine the number of unshared pairs at each designated atom.Be sure your answers are consistent with the formal charges on the formulas.arrow_forwardAssuming the octet rule is obeyed, how many covalent bonds will an oxygen atom form to give a formal charge of zeroarrow_forwardLewis structures showing all valence electrons. Assign formal charges in structure as appropriatearrow_forward
- Draw the Lewis structure of the missing reactant. Make sure to include lone pairs and non-zero formal charges.arrow_forwardProvide all reasonable resonance structures using curved arrows to show the movement of electrons. If no reasonable resonance structures can be drawn, specify “No Resonance Structures”.arrow_forwardDraw the structure, determine the formal charge of all atoms, determine molecular polarity and draw the resonance structure of the following molecule:arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning



