
Concept explainers
(a)
Interpretation:
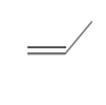
The possibility of cis-trans isomer of Prop-1-ene is to be determined and if it exists, it needs to be drawn.
Concept introduction:
The cis-trans isomers, also known as geometric isomers, are isomers which have same molecular formula but have different permanent position in space.
(b)
Interpretation:
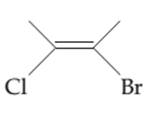
The possibility of cis-trans isomer of 2-Bromo-3-chlorobut-2-ene is to be determined and if it exists then it needs to be drawn.
Concept introduction:
The cis-trans isomers, also known as geometric isomers, are isomers which have same molecular formula but have different permanent position in space.
(c)
Interpretation:
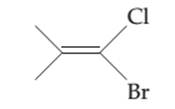
The possibility of cis-trans isomer of 1-Bromo-1-chloro-2-methylpropene is to be determined and if it exists then it needs to be drawn.
Concept introduction:
The cis-trans isomers, also known as geometric isomers, are isomers which have same molecular formula but have different permanent position in space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
CHEMISTRY-TEXT
- Do you predict the product to be the cis isomer, the trans isomer, or a mixture of cis and trans isomers? Explain.arrow_forwardWhich of the ff. molecules can exist as cis and trans isomer?arrow_forwardBased from the figures below, which of the following statements is correct?I. A is an skeletal isomer of D while B and C are positional isomers.II. B and C are alkynes with six Carbons but their structures are different.III. All of the above have the possibility of a cis-trans isomerism.arrow_forward
- a. Which of the following compounds can exist as cis–trans isomers? b. For those compounds that can exist as cis and trans isomers, draw and label the isomers. 1. CH3CH=CHCH2CH2CH3 2. CH3CH2C-CH2CH3=CHCH3 3. CH3CH CHCH3 4. CH3CH2CH CH2arrow_forwardWhich of the following can exist as cis/trans isomers. For any that can write both the cis and the trans structures. (a) CH2=CHCH2CH3 (b) CH3CH2CH=CHClarrow_forwardReplace one atom in CH3CH2Br by another Br atom to form a model of a compound with the formula C2H4Br2. Draw projection drawings and perspective drawings of all possible isomers. Does replacing different hydrogen atoms in CH3CH2Br to produce C2H4Br2 give different isomers? (i.e. Are all the hydrogen atoms equivalent?) If there are multiple isomers, what type of isomerism do they represent?arrow_forward
- Which of the following compounds can exist as cis-trans isomers? b) For those compounds that can exist as cis and trans isomers, draw and label the isomers. 1. CH3CH=CHCH2CH2CH3 2. CH3CH2CCH2CH3=CHCH3 3. CH3CH=CHCH3 4. CH3CH2CH=CH2arrow_forwardProjection drawings of all isomers of C6H4Cl2. Can you convert one into another without breaking any bonds? What type of isomerism do these represent?arrow_forwardDraw possible strucutres for the formula C4H6Cl2, identify the cis and trans isomerdarrow_forward
- What kind of constitutional isomerism do the compounds have?arrow_forwardDirections: Identify what type of structural isomerism is being exhibited by each pairof compounds. Choose from the three most common types of isomerism (e.g. chain,position and functional isomerism).arrow_forwardAre the following two compounds structural isomers, cis-trans isomers, or not isomers?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
