
Concept explainers
(a)
Interpretation:
Thesimplified orbital overlap picture needs to be drawnto indicate the p system and localized lone pairs of the given molecule.
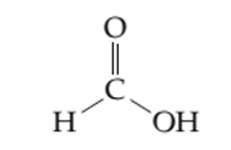
Concept introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In
(b)
Interpretation:
The localized lone pairs and delocalized lone pairs in the given molecule needs to be determined.
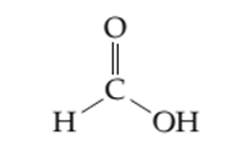
Concept introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
(c)
Interpretation:
Theresonance structure of the given molecule needs to be determined.
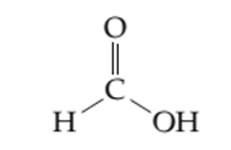
Concept introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
CHEMISTRY-TEXT
- Which of the following molecules contains a nitrogen atom with linear geometry?arrow_forwardIn what type of orbital does each lone pair reside?arrow_forwardWhich of the following molecular formulas are correctly represented by line-bond structures that are consistent with valence rules?arrow_forward
- Differentiate between covalent bonds and generallyweaker interactions such as polar covalent, hydrogen, andionic bonds.arrow_forwardSketch an orbital picture of ethylene, H2C = CH2, and highlight the orbitals that overlap to formeach σ and π bond. Identify the orbitals from each atom that overlap to form the σ and π bonds. Why isthe C⎯ C π bond weaker than the C⎯ C σ bond? Use orbital structure as part of your explanation.arrow_forwardthe actual molecule of which this is a Lewis stricture, which of the labeled distances can change?arrow_forward
- What is the hybridization of the carbon atoms with arrows in each of the following molecules?arrow_forwardGive an example of a molecule containing both aσ- and a π-bond and indicate the two types ofbonds in your molecule.arrow_forwardTotal number of lone pairs of electrons in XeOF4 is ?arrow_forward
- From models of SF4, BrF3, and XeF4, deduce whether differentatom arrangements, called geometrical isomers, are possible, and,if so, sketch them below.Indicate the preferred geometry foreach case and suggest a reason for your choice.Indicatewhich structures have dipole moments, and show their direction. Molecule Dipole Moment Preferred Geometry SF4 BrF3 XeF4arrow_forwardBenzene is commonly regarded as a resonance hybrid ofthe two Kekule structures. but other possible structures canalso contribute. Draw three other structures in which thereare only covalent n bonds (al lowing for bonding betweensome non-adjacent C atoms) and two structures in whichthere is one ionic bond. Why may these structures be ignoredin simple descriptions of the molecule?arrow_forwardCan you solve this (a) Using the language of valence bond theory and orbital energy diagrams, describe the formation and type of bond in the hydroxide ion … WITHOUT considering promotion or hybridization. (b) Draw the Lewis structure of the hydroxide ion and determine the formal charge on both atoms.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning




