
Concept explainers
Interpretation:
Comparing to diphenylamine
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
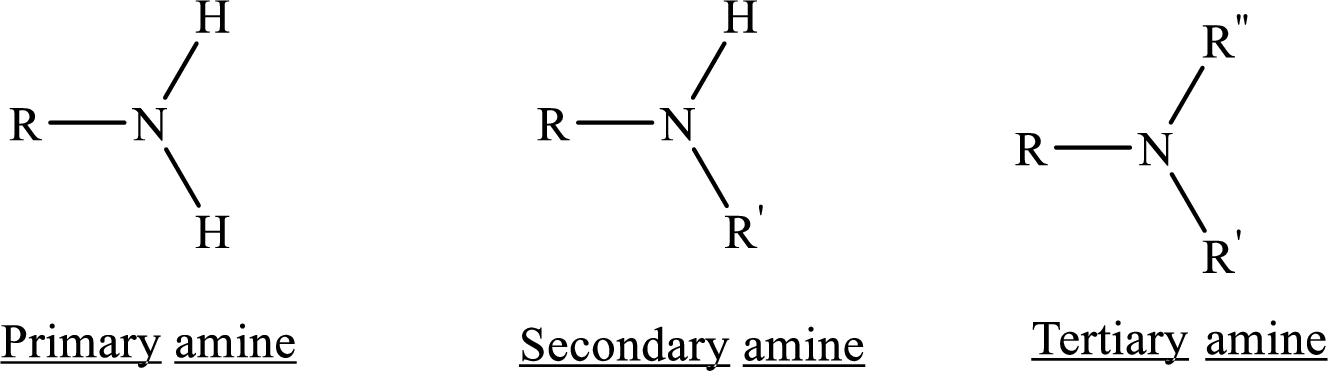
If nitrogen atom of amine group is attached only to alkyl groups, then it is an aliphatic amine.
If nitrogen atom of amine group is attached to at least one aryl groups, then it is an
Lone pair present on nitrogen of aromatic amine takes part in resonance by interacting with the
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- Using pKa Values to Determine Relative Acidity and Basicity Rank the following compounds in order of increasing acidity, and then rank their conjugate bases in order of increasing basicity.arrow_forward4-Methylphenol is more acidic than ethanol (pKa 10.36 vs 16.0) , even though both contain an OH group and a methyl group. Draw the structures of the anions formed from loss of the alcoholic protons from both compounds. Use resonance to explain the difference in their respective acidities.arrow_forwardDetermine the hybridization around the N atom in each amine, andexplain why cyclohexanamine is 106 times more basic than aniline.arrow_forward
- (c) Arrange the following compounds in order of increasing acidity, and explain the reasons for your choice of order: phenol, cyclohexanol, 2-fluorocyclohexanol, 2-fluorophenol.arrow_forwardAt pH 3.0, what percentage (in %) of lactic acid is in its deprotonated form? Hint: The pka of lactic acid is 3.86.arrow_forwardProvide an explanation without using the pka values : Why is phenol stronger acid than butanoic acid?arrow_forward
- 4. The pk, of vanillin is about 9, which is much more acidic than a normal alcohol. Draw a reaction showing the deprotonation of vanillin with NaOH, and then draw six resonance structures of the conjugate base. Draw the hybrid structure and clearly indicate how the negative charge is distributed in the compound.arrow_forwardPhthalic acid and isophthalic acid have protons on two carboxy groups that can be removed with base. (a) Explain why the pKa for loss of the first proton (pKa1) is lower for phthalic acid than isophthalic acid. (b) Explain why the pKa for loss of the second proton (pKa2) is higher for phthalic acid than isophthalic acid.arrow_forwardThe pka for 2-hydroxybenzoic acid (i.e. salicylic acid, used in pain relief and a precursor for aspirin) is 2.98, while the pKa for 3-hydroxybenzoic acid (found in castoreum, a scent- marking substance secreted by beavers) is 4.08. What does this difference in pK₂ tell us about the relative acidities of these two compounds?, Why is there such a significant difference? OH OH 2-hydroxybenzoic acid (pKa = 2.98) OH OH 3-hydroxybenzoic acid (pKa 4.08)arrow_forward
- но HO но он The pK, of ascorbic acid (vitamin C) is 4.17, showing that it is slightly more acidic than acetic acid (CH3CO0H, pKa 4.74). (a) Show the fou r different conjugate bases that would be formed by deprotonation of the four different OH groups in ascorbic acid. (b) Compare the stabilities of these four conjugate bases, and predict which OH group of ascorbic acid is the most acidic. (c) Compare the most stable conjugate base of ascorbic acid with the conjugate base of acetic acid, and suggest why these two compounds have similar acidities, even though ascorbic acid lacks the carboxylic acid (COOH) group.arrow_forwardWrite the equilibrium-constant expressions and obtainnumerical values for each constant in. (a) the basic dissociation of aniline, C6H5NH2. (b) the acidic dissociation of hypochlorous acid,HClO. (c) the acidic dissociation of methyl ammoniumhydrochloride, CH3NH3Cl. (d) the basic dissociation of NaNO2. (e) the dissociation of H3AsO3to H3O+and AsO33-. (f) the reaction of C2O42-with H2O to give H2C2O4and OH-. show solutionarrow_forwardChemical Equilibrium Write the equilibrium-constant expressions and obtain numerical values for each constant in (a) the basic dissociation of aniline, C6H5NH2. (b) the acidic dissociation of hypochlorous acid, HClO. (c) the acidic dissociation of methyl ammonium hydrochloride, CH3NH3Cl. (d) the basic dissociation of NaNO2. (e) the dissociation of H3AsO3 to H3O+ and AsO33- Using step-by-step processarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

