
Organic Chemistry
8th Edition
ISBN: 9781337516402
Author: Brown
Publisher: Cengage
expand_more
expand_more
format_list_bulleted
Question
Chapter 23, Problem 23.52P
Interpretation Introduction
Interpretation:
The synthesis of N-methylmorpholine has to be shown.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid.
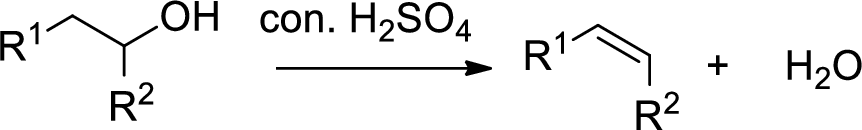
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What carboxylic acid and amine are needed to synthesize the pain reliever phenacetin? Phenacetin was once a component of the over-the-counter pain reliever APC (aspirin, phenacetin, caffeine), but it is no longer used because of its kidney toxicity.
Define the simplest method to synthesize an amine ?
Aliphatic amines are more basic than ammonia, whereas aromatic amines are less basic than ammonia.
Is that true or false?
Chapter 23 Solutions
Organic Chemistry
Ch. 23.1 - Prob. 23.1PCh. 23.2 - Prob. 23.2PCh. 23.2 - Prob. 23.3PCh. 23.2 - Prob. 23.4PCh. 23.5 - Prob. 23.5PCh. 23.5 - Prob. AQCh. 23.5 - What is the hybridization of the nitrogen in...Ch. 23.5 - Prob. CQCh. 23.5 - The pKas of the conjugate acids of aniline and...Ch. 23.5 - Prob. EQ
Ch. 23.5 - Prob. FQCh. 23.5 - Prob. GQCh. 23.5 - Select the stronger acid from each pair of...Ch. 23.6 - Prob. 23.7PCh. 23.6 - Prob. 23.8PCh. 23.6 - Prob. 23.9PCh. 23.7 - Prob. 23.10PCh. 23.8 - Prob. 23.11PCh. 23.8 - Prob. 23.12PCh. 23.8 - Prob. 23.13PCh. 23.9 - Prob. 23.14PCh. 23.10 - In Example 23.15, you considered the product of...Ch. 23 - Prob. 23.16PCh. 23 - Prob. 23.17PCh. 23 - Prob. 23.18PCh. 23 - Prob. 23.19PCh. 23 - Prob. 23.20PCh. 23 - Prob. 23.21PCh. 23 - Prob. 23.22PCh. 23 - Account for the formation of the base peaks in...Ch. 23 - Prob. 23.24PCh. 23 - Select the stronger base from each pair of...Ch. 23 - The pKa, of the conjugate acid of morpholine is...Ch. 23 - Which of the two nitrogens in pyridoxamine (a form...Ch. 23 - Prob. 23.28PCh. 23 - Prob. 23.29PCh. 23 - Prob. 23.30PCh. 23 - Prob. 23.31PCh. 23 - Suppose you have a mixture of these three...Ch. 23 - Prob. 23.33PCh. 23 - Prob. 23.34PCh. 23 - Prob. 23.35PCh. 23 - Prob. 23.36PCh. 23 - Prob. 23.37PCh. 23 - (S)-Glutamic acid is one of the 20 amino acid...Ch. 23 - Prob. 23.39PCh. 23 - Propose a structural formula for the compound...Ch. 23 - Prob. 23.41PCh. 23 - The pyrolysis of acetic esters to give an alkene...Ch. 23 - Propose steps for the following conversions using...Ch. 23 - Show how to bring about each step in this...Ch. 23 - Show how to bring about each step in the following...Ch. 23 - Prob. 23.48PCh. 23 - Prob. 23.49PCh. 23 - Methylparaben is used as a preservative in foods,...Ch. 23 - Prob. 23.51PCh. 23 - Prob. 23.52PCh. 23 - Propose a synthesis for the systemic agricultural...Ch. 23 - Prob. 23.54PCh. 23 - Several diamines are building blocks for the...Ch. 23 - Prob. 23.56PCh. 23 - Prob. 23.57PCh. 23 - Prob. 23.58PCh. 23 - Prob. 23.59PCh. 23 - Following is a retrosynthesis for the coronary...Ch. 23 - Prob. 23.61PCh. 23 - Prob. 23.62PCh. 23 - Given this retrosynthetic analysis, propose a...Ch. 23 - Prob. 23.64PCh. 23 - Following is a series of anorexics (appetite...Ch. 23 - Prob. 23.66PCh. 23 - Prob. 23.67PCh. 23 - Show how the synthetic scheme developed in Problem...Ch. 23 - Prob. 23.69PCh. 23 - Prob. 23.70PCh. 23 - Prob. 23.71PCh. 23 - Prob. 23.72P
Knowledge Booster
Similar questions
- 6. The reaction between aniline and nitrous acid at low temperature yields A) an N-nitroso amine B) a diazonium salt C) a nitrile D) an amine nitrite salt 7. An organic nitrogen compound, X, gives ammonia on warming with dilute aqueous sodium hydroxide, X could be A) ethanamide B) ethylamine C) phenylamine D) amino ethanoic acidarrow_forwardDraw the struetre of the following three isomeic amides with chemical feruula CHNO. Amide #1: (E)-N,N-dimethyl-2-butenamide Amide #2: (7)-N-methyl-3-pentenamide Amide #3: (7)-3-hexenamide C'onsider Z sterenchemistry of alkenes. • Draw one structure per sketcher. Add additional sketchers asing the drop-down memu in the hottom right comer. Sepamte structares with - signs from the drop-down mena. O00. IF ChemDoodle ChemDoodie ChemDoodle just the first one is what I could really use help on Amide #1: (E)-N,N-dimethyl-2-butenamidearrow_forwardThe following three derivatives of succinimide are anticonvulsants and have found use in the treatment of epilepsy, particularly petit mal seizures. Ph Ph `N' `N' ČH3 ČH3 Methsuximide Ethosuximide Phensuximide Following is a synthesis of phensuximide. CN Ph CN Ph CN 1. NaOH, H2O 2. HC, Н20 NaOEt KCN Ph-CHO cOOEt H cOOEt NC COOEt 3. Нeat Ethyl cyanoacetate (A) (B) Benzaldehyde Ph Ph Ph CH;NH2 НООС СООН Et0oC COOEt `N' (C) (D) ČH3 Phensuximide Methsuximide is formed by a similar pathway to that shown for phensuximide. Draw the structure of the compound that reacts with ethyl cyanoacetate in the synthesis of methsuximide.arrow_forward
- Show how to synthesize the following amines from the indicated starting materials below. Each of the final products may require several sequential steps. Draw the structure for each of the steps and show the reagents required. Starting material Final product b) Benzene m-bromoaniline c) ethylbenzene p-ethylaniline + o-ethylaniline d) benzene p-bromoaniline + o-bromoanilinearrow_forward+ Amines can be prepared by converting other functional groups. Classify these reactions and choose the correct reagents. NO₂ NH₂ Three reactions that can be used to prepare amines are shown below. NH₂ NH₂ NH₂ Classify these reactions. A) Elimination B) Substitution C) Reduction D) Oxidation E) Proton transfer reactionsarrow_forwardIn the mid-1930s a substance was isolated from a fungus that is a parasite of ryes and other grasses. This alkaloid, lysergic acid, has been of great interest to chemists because of its strange, dramatic action on the human mind. Many derivatives of lysergic acid are known, some with medicinal applications. Perhaps the best known derivative of lysergic acid is the potent hallucinogen lysergic acid diethylamide (LSD): మగవా జి N-H LSD (CH25N;O) Like other alkaloids, LSD is a weak base, with Kp = 7.6 × 107. What is the pH of a 0.94 M solution of LSD? pH =arrow_forward
- Indicate whether the following statement is true or false. Aliphatic amines are more basic than ammonia, whereas aromatic amines are less basic than ammonia. When amines are reacted with bases, they form ammonium salts. Benzenesulfonyl chloride or p-toluenesulfonylchloride give N-substituted sulfonamides with primary and secondary amines. Derivative of primary amines is insoluble in dilute NaOHarrow_forward16-13 Classify each amino group as primary, secondary, or tertiary and as aliphatic or aromatic. Serotonin (a neurotransmitter) Diphenhydramine (the hydrochloride salt is the antihistamine Benadryl) Lysine (an amino acid)arrow_forward16-26 The p/fb of amphetamine is approximately 3.2 Amphetamine (a) Which form of amphetamine (the free base or its conjugate acid) would you expect to be present at pH 1.0, the pH of stomach acid? (b ) Which form of amphetamine would you expect to be present at pH 7.40, the pH of blood plasma?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning