
Concept explainers
(a)
Interpretation:
The reagent and condition has to be proposed for step 1,2,3 and 5.
Concept introduction:
Hydrogenolysis:
Nitration: The formation of nitro group in a
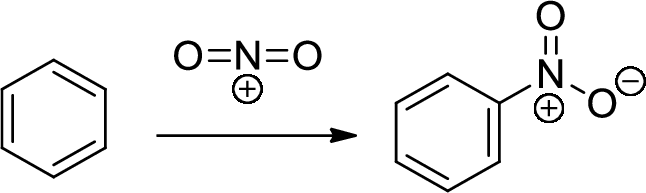
(b)
Interpretation:
The mechanism is to be proposed for iodination of 3-aminobenzoic acid.
Concept introduction:
Electrophilic
Halogenations on benzene:
Halogenation is one of the electrophilic substitution reactions. Halogens reaction with benzene (or electron donating group present in the benzene ring) which gives the corresponding halogenated compound.
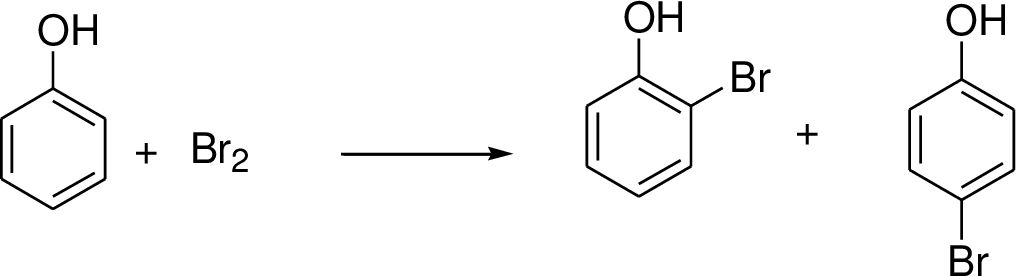
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- Estrogens are female sex hormones, the most potent of which is B-estradiol. OH AYA Но B-Estradiol In recent years, chemists have focused on designing and synthesizing molecules that bind to estrogen receptors. One target of this research has been nonsteroidal estrogen antagonists, compounds that interact with estrogen receptors and block the effects of both endogenous and exogenous estrogens. A feature common to one type of nonste- roidal estrogen antagonist is the presence of a 1,2-diphenylethylene with one of the benzene rings bearing a dialkylaminoethoxyl substituent. The first nonsteroidal estrogen antagonist of this type to achieve clinical importance was tamoxifen, now an important drug in the treatment of breast cancer. Tamoxifen has the Z configuration shown here. NMeg A B NMeg ? ОН Tamoxifen Propose reagents for the conversion of A to tamoxifen. Note: The final step in this synthesis gives a mixture of E and Z isomers.arrow_forwardWhat are the derivate reagents for the following compound: MDMA (N-methyl-N-trifluoroacetyl-3,4-methylenedioxyamphetamine)?arrow_forwardSeveral diamines are building blocks for the synthesis of pharmaceuticals and agro-chemicals. Show how both 1,3-propanediamine and 1,4-butanediamine can be prepared from acrylonitrile.arrow_forward
- How would you use IR and ¹H-NMR spectroscopy to distinguish between these two isomeric compounds? For IR, consider the absorption of the carbonyl group. For ¹H-NMR, consider the number of signals observed. Select the IR frequency and number of NMR signals that is diagnostic for either isomer a or isomer b. Isomer A: cyclopentanecarboxamide Isomer B: 6-hydroxyhexanenitrile IR: ¹H-NMR: cm-1. a-4 a-5 b-3 b-4 b-5 b-6 signals a-1680 a-1690 a-1735 a-1810 a-2250 b-1650 b-1690 b-1710 b-2250 24arrow_forwardSuggest a possible structure for Compound X.arrow_forwardWhy do you wash the dichloromethane solution of your reductive amination product with sodium bicarbonate, rather than dilute aqueous HCl? a) Sodium bicarbonate is a good method of removing aldehydes from organic solvent.b) The amine product will be protonated by acid and remain in the aqueous layer as a salt.c) Sodium bicarbonate transfers the amine starting material into the aqueous layer.d) Sodium bicarbonate reacts with leftover NaBH(OAc)3 and removes it from the mixture.arrow_forward
- Aspirin is the common name for the compound acetylsalicylic acid, widely used as a fever reducer and as a pain killer. Salicylic acid, whose name comes from Salix, the willow family of plants, was derived from willow bark extracts. In folk medicine, willow bark teas were used as headache remedies and other tonics. Nowadays, salicylic acid is administered in the form of aspirin which is less irritating to the stomach than salicylic acid. To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride. A small amount of a strong acid is used as a catalyst which speeds up the reaction. In this experiment, phosphoric acid will be used as the catalyst. The excess acetic acid will be quenched with the addition of water. The aspirin product is not very soluble in water so the aspirin product will precipitate when water is added. The synthesis reaction of aspirin is shown below: Actic anhydride 5 ml. Acetic acid Salicylic acid 28 Acetylsalicylie acid Procedure 1) Mix salicylic…arrow_forwardEistrogens are female sex hormones, the most potent of which is B-estradiol. OH H,C но B-Estradiol In recent years, chemists have focused on designing and synthesizing molecules that bind to estrogen receptors. One target of this research has been nonsteroidal estrogen antagonists, compounds that interact with estrogen receptors and block the effects of both endogenous and exogenous estrogens. A feature common to one type of nonste- roidal estrogen antagonist is the presence of a 1,2-diphenylethylene with one of the benzene rings bearing a dialkylaminoethoxyl substituent. The first nonsteroidal estrogen antagonist of this type to achieve clinical importance was tamoxifen, now an important drug in the treatment of breast cancer. Tamoxifen has the Z configuration shown here. но NMe, NMeg ? B NMeg ? "NMe, ОН Tamoxifen Propose reagents for the conversion of A to tamoxifen. Note: The final step in this synthesis gives a mixture of E and Z isomers.arrow_forwardHow harmful Terephthalic acid is paarticularly in human health and environment?arrow_forward
- In preparation of dibenzalacetone from benzaldehyde: Give two techniques that can be used to purify partially air-oxidized benzaldehyde. Indicate how each technique works in purifying benzaldebyde from its air-oxidized impurities.arrow_forward5 The compounds labeled benzophenone-3 (CH,O,) and benzophenone-5 (CHNAO,S) are found in certain sunscreens. Would you expect a sunscreen made with benzophenone-3 or benzophenone-5 to be more waterproof? Explain your choice.arrow_forwardIdentify the amine and coupling components of this azo dye. O₂N Narrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning



