
Concept explainers
(a)
Interpretation: To characterize the change of citrate to isocitrate in terms of carbon atom as (1) a C6 to C6 change, (2) a C6 to C5 change, (3) a C5 to C4 change, or (4) a C4 to C4 change.
Concept introduction: Citric acid cycle is the third stage of the biochemical energy production process. The cycle includes the reactions in which the acetyl part of acetyl CoA is oxidized and leads to the formation of carbon dioxide and
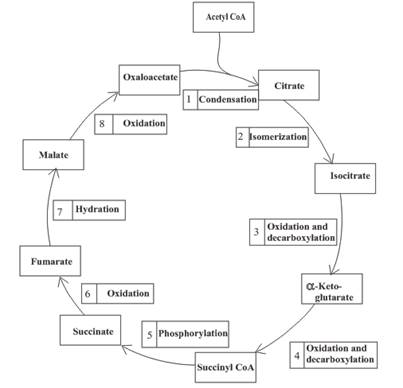
The citric acid cycle is included in the common
(b)
Interpretation: To characterize the change of succinate to fumarate in terms of carbon atom as (1) a C6 to C6 change, (2) a C6 to C5 change, (3) a C5 to C4 change, or (4) a C4 to C4 change.
Concept introduction: Citric acid cycle is the third stage of the biochemical energy production process. The cycle includes the reactions in which the acetyl part of acetyl CoA is oxidized and leads to the formation of carbon dioxide and
An overview of the citric acid cycle is as follows:
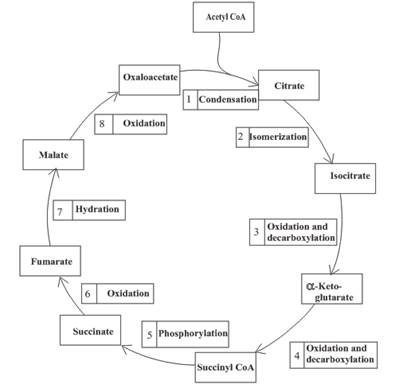
The citric acid cycle is included in the common metabolic pathway because the reactions in this stage are the same for different kinds of food.
(c)
Interpretation: To characterize the change of malate to oxaloacetate in terms of carbon atom as (1) a C6 to C6 change, (2) a C6 to C5 change, (3) a C5 to C4 change, or (4) a C4 to C4 change.
Concept introduction: Citric acid cycle is the third stage of the biochemical energy production process. The cycle includes the reactions in which the acetyl part of acetyl CoA is oxidized and leads to the formation of carbon dioxide and
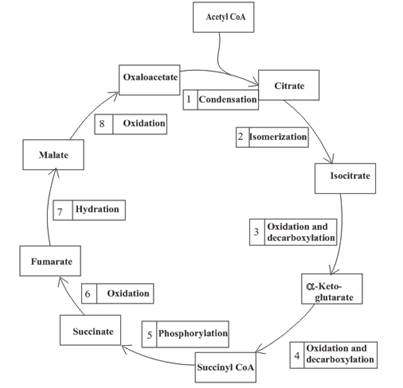
The citric acid cycle is included in the common metabolic pathway because the reactions in this stage are the same for different kinds of food.
(d)
Interpretation: To characterize the change of isocitrate to
Concept introduction: Citric acid cycle is the third stage of the biochemical energy production process. The cycle includes the reactions in which the acetyl part of acetyl CoA is oxidized and leads to the formation of carbon dioxide and

The citric acid cycle is included in the common metabolic pathway because the reactions in this stage are the same for different kinds of food.
Trending nowThis is a popular solution!

Chapter 23 Solutions
GENERAL,ORGANIC,+BIO.CHEM.-MINDTAP
- Which of these is associated with production of ATP during glycolysis? (Select all that apply) a) Conversion of 1,3-BPG to 3PG b) Conversion of 1,3-BPG to 2,3-BPG c) Conversion of PEP to pyruvate d) Conversion of 2,3-BPG to 2PG e) Phosphorylation of glucosearrow_forwardIf 14CO2 (radioactive carbon) were incorporated into the TCA cycle via the Pyruvate Carboxylase reaction which of the following molecules would contain radioactive carbon? a. Oxaloacetate b. Citric Acid c. Isocitrate d. Alpha-ketoglutarate e. Succinyl-CoA f .Succinate g. Fumarate h. Malatearrow_forwardAnswer the following questions below: 1. Individuals with a thiamine-deficient diet have relatively high levels of pyruvate in their blood. Explain in biochemical terms. 2. How would a riboflavin deficiency affect the functioning of the citric acid cycle? Explain your answer.arrow_forward
- Step 6 of the citric acid cycle is shown. Which statement best describes the role of FAD in this reaction? CH₂ lal CH₂ FAD FADH₂ CO₂ CO₂ succinate CH CH fumarate OA) FAD causes the oxidation of succinate to form fumarate. B) FAD causes succinate to undergo hydration to form fumarate. C) FAD causes the isomerication of succinate to form fumarate. D) FAD causes the reduction of succinate to form fumarate.arrow_forwardConsider the fatty acids: (a) Arachidic acid (C20H40O2); molar mass = 312.5 g/mol) (b) Palmitoleic acid(C16H30O2); molar mass = 256.4 g/mol). i. How many cycles of β -oxidation are needed for complete oxidation?ii. How many molecules of acetyl CoA are formed from its complete catabolism?iii. Calculate the number of molecules (moles) of ATP formed (net) by the completecatabolism of each fatty acid (show your calculation).iv. Calculate number of moles of ATP formed per gram of each fatty acid metabolized.arrow_forwardThe citric acid cycle enzyme at which FADH2 is generated is: A) isocitrate dehydrogenase. B) x-ketoglutarate dehydrogenase. C) succinate dehydrogenase. OD) malate dehydrogenase.arrow_forward
- Identify the following: a. An oxidative process that releases energy b. Cellular localization of the electron transport chain during cellular respiration. c. The number of double bonds present in the metabolic precursor of all prostaglandins.arrow_forwardGlucosamine is a common compound that inhibits hexokinase action (the transfer of a phosphate group to glucose during glycolysis). A) Describe the consequences of glucosamine exposure on cellular respiration. B) Is glucosamine exposure fatal? Why or why not?arrow_forwardDEFINE THE FOLLOWING TERMS: a) Gluconeogenesis b) Glycolysis c) Citric Acid Cycle Please explain this in-detailed and longer, thank you.arrow_forward
- identify the reactions of the citric acid cycle that provide reducing power for the electron-transport chain a. conversion of succinyl-CoA to succinate b. conversion of fumarate to malate c. conversion of succinate to fumarate d. conversion of malate to oxaloacetatearrow_forwardConsider the following statements: (1) the products of the first two stages of cholesterol biosynthesis are, respectively, mevalonate (C6) and isopentyl pyrophosphate (2) Over 90 percent of the total dietary lipids are triaglycerols (3) The activation stages of fatty acid oxidation involves both CoA & ATP Choices: a. Only one of the statements is true b. All three statements are true c. Two of the three statements are true d. None of the statements are truearrow_forwardThe first reaction in the citric acid cycle is binding * OA) carbon dioxide to a four-carbon (C4) molecule. OB) carbon dioxide to a five-carbon (C5) molecule. C) acetyl-CoA to a C4 molecule. D) acetyl-CoA to a C5 molecule. E) acetyl-CoA to citric acid What are the functions of dietary fat? *arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





