
Concept explainers
a)
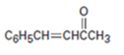
Interpretation:
Whether the compound shown can be prepared by a mixed aldol reaction is to be stated. Further the reactants from which it can be prepared are to be given.
Concept introduction:
Mixed aldol condensation leads to a single product if i) one of the carbonyl partners contains no α hydrogen atoms but contains an unhindered carbonyl group and ii) if one of the carbonyl partners are much more acidic than the other and thus forms an enolate anion in preference to other.
To state:
Whether the compound shown can be prepared by a mixed aldol reaction.
To give:
The reactants from which it can be prepared.
b)
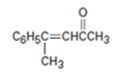
Interpretation:
Whether the compound shown can be prepared by a mixed aldol reaction is to be stated. Further the reactants from which it can be prepared are to be given.
Concept introduction:
Mixed aldol condensation leads to a single product if i) one of the carbonyl partners contains no α hydrogen atoms but contains an unhindered carbonyl group and ii) if one of the carbonyl partners are much more acidic than the other and thus forms an enolate anion in preference to other.
To state:
Whether the compound shown can be prepared by a mixed aldol reaction.
To give:
The reactants from which it can be prepared.
c)
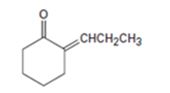
Interpretation:
Whether the compound shown can be prepared by a mixed aldol reaction is to be stated. Further the reactants from which it can be prepared are to be given.
Concept introduction:
Mixed aldol condensation leads to a single product if i) one of the carbonyl partners contains no α hydrogen atoms but contains an unhindered carbonyl group and ii) if one of the carbonyl partners are much more acidic than the other and thus forms an enolate anion in preference to other.
To state:
Whether the compound shown can be prepared by a mixed aldol reaction.
To give:
The reactants from which it can be prepared.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- Give the structures of the aldol products that form when each of the following compounds or mixtures is treated with NaOH. The top three compounds are aldol dimerizations and require 2 moles of the reactant listed, while the two compounds on the bottom row are being used in a mixed aldol reaction. Butanal, Cyclopentanone, and Acetophenone (PhCOCH3) p-chlorobenzaldehyde and 2,2-dimethylcyclohexanonearrow_forwardA student tried to prepare the following compounds using aldol condensations. Which of these compounds was she successful in synthesizing? Explain why the other syntheses were not successful.arrow_forwardWhat are the products of an aldol cyclization for the following compound?arrow_forward
- Which of the following aldehydes does not undergo an aldol condensation when mixed with base? A. (CH3)3CCHO B. C6H5CH2CHO C. CH3CHO D. CH3CH2CHOarrow_forwardWhat starting materials do you need to synthesize each of the following compounds using an intramolecular aldol reaction?arrow_forwardHow each of the following could be synthesized by way of aldol condensation?a. n-butyl alcohol b. 4-methyl,2-pentanolarrow_forward
- What aldol product is formed when two molecules of butanal react together in the presence of base? What reagents are needed to convert this product to each of the following compounds?arrow_forwardPredict the major products of the following base-catalyzed aldol condensations withdehydration. ) 2,2@dimethylpropanal + acetophenonearrow_forwardPredict the major products of the following base-catalyzed aldol condensations withdehydration.(a) benzophenone (PhCOPh) + propionaldehyde(b) 2,2@dimethylpropanal + acetophenonearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT



