
Concept explainers
a)
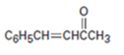
Interpretation:
Whether the compound shown can be prepared by a mixed aldol reaction is to be stated. Further the reactants from which it can be prepared are to be given.
Concept introduction:
Mixed aldol condensation leads to a single product if i) one of the carbonyl partners contains no α hydrogen atoms but contains an unhindered carbonyl group and ii) if one of the carbonyl partners are much more acidic than the other and thus forms an enolate anion in preference to other.
To state:
Whether the compound shown can be prepared by a mixed aldol reaction.
To give:
The reactants from which it can be prepared.
b)
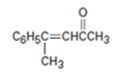
Interpretation:
Whether the compound shown can be prepared by a mixed aldol reaction is to be stated. Further the reactants from which it can be prepared are to be given.
Concept introduction:
Mixed aldol condensation leads to a single product if i) one of the carbonyl partners contains no α hydrogen atoms but contains an unhindered carbonyl group and ii) if one of the carbonyl partners are much more acidic than the other and thus forms an enolate anion in preference to other.
To state:
Whether the compound shown can be prepared by a mixed aldol reaction.
To give:
The reactants from which it can be prepared.
c)
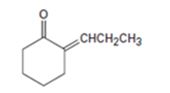
Interpretation:
Whether the compound shown can be prepared by a mixed aldol reaction is to be stated. Further the reactants from which it can be prepared are to be given.
Concept introduction:
Mixed aldol condensation leads to a single product if i) one of the carbonyl partners contains no α hydrogen atoms but contains an unhindered carbonyl group and ii) if one of the carbonyl partners are much more acidic than the other and thus forms an enolate anion in preference to other.
To state:
Whether the compound shown can be prepared by a mixed aldol reaction.
To give:
The reactants from which it can be prepared.
Trending nowThis is a popular solution!

Chapter 23 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- How can the following compounds be prepared from a carbonyl compound that has one less carbon than the desired product?arrow_forwardDraw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2arrow_forwardShow how the following products can be synthesized via a Michael reaction.arrow_forward
- Identify the reactant that you would use to make the following compound via an aldol addition reaction. ? NaOH, H₂O H H in we H ya H HOarrow_forwardThe following synthesis requires more than one step. Specify the reagents you would use to carry it out.arrow_forwardA student tried to prepare the following compounds using aldol condensations. Which of these compounds was she successful in synthesizing? Explain why the other syntheses were not successful.arrow_forward
- What are the products of an aldol cyclization for the following compound?arrow_forwardShow two reaction schemes to synthesize the following compounds. Pick one reactionscheme and draw the step-by-step reaction mechanismarrow_forwardAcetic acid has been mixed with isoamyl alcohol to produce isoamyl acetate giving off a banana smell. Propose a reaction mechanism for this reaction.arrow_forward
- Draw the structures A, B and C for the reaction sequence below. Dehydration via an E1cb mechanism Michael Reaction Intramolecular Aldol Reaction A В КОН /ETOH / heat C КОН /ETOH КОН /ETOHarrow_forwardWhat reagents are required to carry out the following synthesis?arrow_forwardChemistry Questionarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





