
Concept explainers
(a)
Interpretation:
Number of carbon-carbon sigma bonds present in the given 2-butyne has to be identified.
Concept introduction:
Sigma bonds: A sigma bond is a covalent bond formed by head on overlap of atomic orbitals that carries two electrons. The bonding represented as a single line between two atoms.
Head-on overlap:
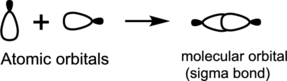
Head-on overlap of two atomic orbitals forms a molecular orbital known as sigma bonds.
Side-to-side overlap:
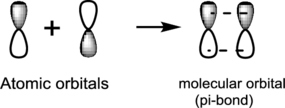
Side-to-side overlap of two atomic orbitals forms a molecular orbital known as pi-bonds.
(b)
Interpretation:
Number of carbon-carbon sigma bonds present in the given anthracene has to be identified.
Concept introduction:
Refer part ‘a’
(c)
Interpretation:
Number of carbon-carbon sigma bonds present in the given 2,3-dimethylpentane has to be identified.
Concept introduction:
Refer part ‘a’
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
Chemistry
- Organic Chemistry HW: 2,6-dimethyloct-2-ene cannot be handwritten, explanation of print the structural formula of your molecule (expanded or condensed) and indicate the functional groups present by highlighting, circling, or color-coding each and labeling the group.arrow_forwardGive the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forwardPlease draw the skeletal isomers of chlorohexane, without those with branched carbon chains.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





