
Concept explainers
Draw the products formed in the crossed aldol reaction of phenylacetaldehyde
(a)
Interpretation: The product that is formed in the given crossed aldol reaction of phenylacetaldehyde
Concept introduction: The crossed aldol reaction takes place between the two different carbonyl compounds that is between two different aldehydes or two different ketones or one aldehyde and one ketone. The crossed aldol reaction takes place only if the carbonyl compound contains an acidic
Answer to Problem 24.8P
The product that is formed in the given crossed aldol reaction of phenylacetaldehyde
![]()
Explanation of Solution
The product that is formed in the given crossed aldol reaction is shown below.
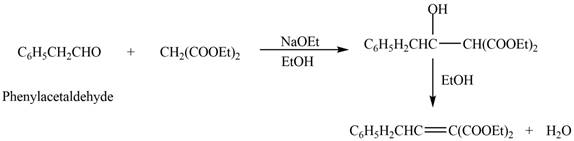
Figure 1
In this crossed aldol reaction, phenylacetaldehyde reacts with
The product that is formed in the given crossed aldol reaction of phenylacetaldehyde
(b)
Interpretation: The product that is formed in the given crossed aldol reaction of phenylacetaldehyde
Concept introduction: The crossed aldol reaction takes place between the two different carbonyl compounds that is between two different aldehydes or two different ketones or one aldehyde and one ketone. The crossed aldol reaction takes place only if the carbonyl compound contains an acidic
Answer to Problem 24.8P
The product that is formed in the given crossed aldol reaction of phenylacetaldehyde
![]()
Explanation of Solution
The product that is formed in the given crossed aldol reaction is shown below.
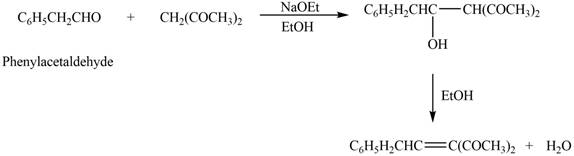
Figure 2
In this crossed aldol reaction, phenylacetaldehyde reacts with
The product that is formed in the given crossed aldol reaction of phenylacetaldehyde
(c)
Interpretation: The product that is formed in the given crossed aldol reaction of phenylacetaldehyde
Concept introduction: The crossed aldol reaction takes place between the two different carbonyl compounds that is between two different aldehydes or two different ketones or one aldehyde and one ketone. The crossed aldol reaction takes place only if the carbonyl compound contains an acidic
Answer to Problem 24.8P
The product that is formed in the given crossed aldol reaction of phenylacetaldehyde
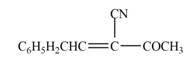
Explanation of Solution
The product that is formed in the given crossed aldol reaction is shown below.
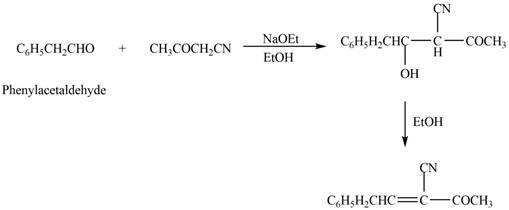
Figure 3
In this crossed aldol reaction, phenylacetaldehyde reacts with
The product that is formed in the given crossed aldol reaction of phenylacetaldehyde
Want to see more full solutions like this?
Chapter 24 Solutions
ORGANIC CHEMISTRY
- Draw the products of the diethyl malonate: (1) sodium ethoxide; (2) isobutyl bromide; (3) HCl, H2O + Δarrow_forward4) The following enone, 3-methylcyclopent-2-enone, was prepared through an intramolecular aldol condensation followed by a dehydration. Draw the starting diketone and the intermediate cyclic ß-hydroxy ketone. 3-methylcyclopent-2-enonearrow_forwardComparing Hydration Products Using Two Different Methods Draw the product formed when CH3CH2C=CH is treated with each of the following sets of reagents: (a) H2O, H2SO4, HgSO4; and (b) R2BH, followed by H2O2, HO−.arrow_forward
- Draw the product formed when phenylacetic acid (C6H5CH2COOH) istreated with following reagent. With some reagents, no reaction occurs. [1] NaOH; [2] CH3COClarrow_forwardDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] HC≡CNa; [2] H2Oarrow_forwardDraw the organic products formed when 2-bromopentan-3-one (CH3CH2COCHBrCH3) is treated with each reagent: (a) Li2CO3, LiBr, DMF; (b) CH3CH2NH2; (c) CH3SHarrow_forward
- Draw the product(s) formed when A is treated with each reagent. (a) NaBH4, CH3OH (b) [1] LiAlH4; [2] H2O (c) CH3CH2MgBr; [2] H2Oarrow_forwardKetoprofen, like ibuprofen, is an anti-inflammatory analgesic. How can ketoprofen be synthesized from the given starting material?arrow_forwardLabel the α and β carbons in attached alkyl halide. Draw all possible elimination products formed when each alkyl halide is treated with K+−OC(CH3)3.arrow_forward
- Draw the products formed when CH3CHC ≡ CCH2CH3 is treated with each reagent: (a) Br2(2 equiv); (b) Cl2 (1 equiv).arrow_forwardDraw the product formed when phenylacetic acid (C6H5CH2COOH) istreated with each reagent. With some reagents, no reaction occurs. NaHCO3arrow_forwardIdentify the lettered compounds in each reaction sequence.Draw the product formed when phenylacetonitrile (C6H5CH2CN) istreated with below reagent. H2O, −OHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





