
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24.14, Problem 18P
Using Mechanism
as a guide, write a stepwise mechanism for the acid hydrolysis of methyl
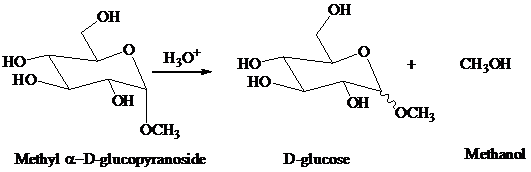
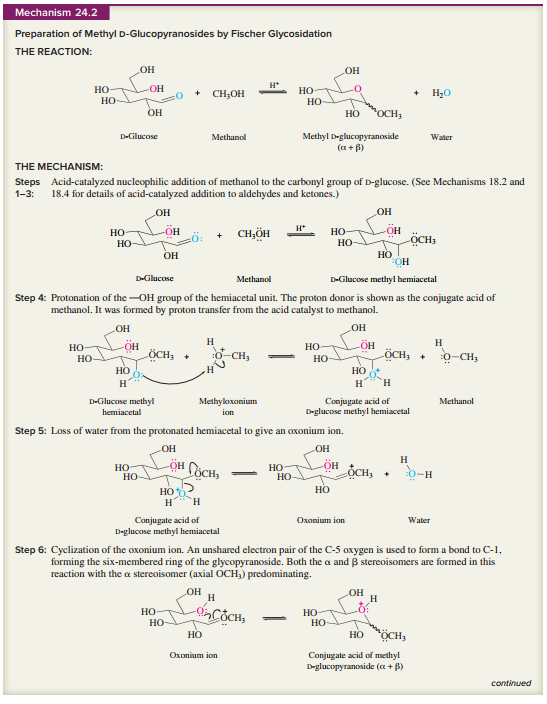
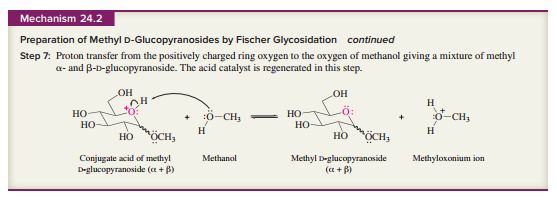
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Show the product that results when fructose is treated with an excess of methyl iodideand silver oxide.(b) Show what happens when the product of part (a) is hydrolyzed using dilute acid.(c) What do the results of parts (a) and (b) imply about the hemiacetal structure of fructose?
1.Explain what is anomeric effect and why it happens?
2.Explain how a glycosyl acetimidate can loss its glycosyl donor activity?
Draw the detailed mechanism of metformin synthesis
Chapter 24 Solutions
Organic Chemistry - Standalone book
Ch. 24.2 - Prob. 1PCh. 24.3 - Problem 24.2 Which aldotetrose in the structure...Ch. 24.4 - Prob. 3PCh. 24.4 - Prob. 4PCh. 24.6 - Prob. 5PCh. 24.6 - Prob. 6PCh. 24.7 - Prob. 7PCh. 24.8 - Prob. 8PCh. 24.8 - Prob. 9PCh. 24.9 - Prob. 10P
Ch. 24.10 - Prob. 11PCh. 24.10 - Prob. 12PCh. 24.11 - Prob. 13PCh. 24.12 - Prob. 14PCh. 24.14 - Prob. 15PCh. 24.14 - Prob. 16PCh. 24.14 - Prob. 17PCh. 24.14 - Using Mechanism 24.2 as a guide, write a stepwise...Ch. 24.15 - Prob. 19PCh. 24.17 - Prob. 20PCh. 24.17 - Prob. 21PCh. 24.18 - Prob. 22PCh. 24.18 - Prob. 23PCh. 24.18 - Prob. 24PCh. 24.20 - Prob. 25PCh. 24 - Prob. 26PCh. 24 - Prob. 27PCh. 24 - Prob. 28PCh. 24 - Prob. 29PCh. 24 - Prob. 30PCh. 24 - Prob. 31PCh. 24 - Prob. 32PCh. 24 - Prob. 33PCh. 24 - Prob. 34PCh. 24 - Prob. 35PCh. 24 - Methyl glycosides of 2-deoxy sugars have been...Ch. 24 - Prob. 37PCh. 24 - Prob. 38PCh. 24 - Prob. 39PCh. 24 - Prob. 40PCh. 24 - Treatment of d-mannose with methanol in the...Ch. 24 - Prob. 42PCh. 24 - Prob. 43PCh. 24 - Prob. 44PCh. 24 - Prob. 45DSPCh. 24 - Prob. 46DSPCh. 24 - Prob. 47DSPCh. 24 - Prob. 48DSPCh. 24 - Prob. 49DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (i) Acetylation of glucose with acetic anhydride gives glucosepentaacetate. Write the structure of the pentaacetate.(ii) Explain why glucose pentaacetate does not react with hydroxylamine?arrow_forwardAs shown in Mechanism 29.2, the nal steps in the Edman degradation result in rearrangement of a thiazolinone to an Nphenylthiohydantoin. Draw a stepwise mechanism for this acid-catalyzed reaction.Mechanism 29.2: Edman Degradationarrow_forwardProvide a procedure for the synthesis of "L-gulono-Lactone" from "D-gluconic acid lactone"arrow_forward
- Illustrate a chemical equation showing the following: 1. Hydrogenation of triarachidonin to Triarachidin in the presence of Nickel 2. Hydrolysis of alpha-stearo-beta-oleoarachidonate 3. Hydrolysis of tricaprin 4. Saponification of tripalmitin with 3 moles NaOH A.M.D.G.arrow_forward1. Predict products of the reaction of Esterification amino acid? 2. Predict products of the reaction of Cylation amino acid? 3. Predict products of the reaction with ninhydrin amino acid?arrow_forwardDraw a stepwise mechanism for the conversion of geranyl diphosphate to α-terpinene. Steroidsarrow_forward
- 4(a) Suggest a test you will use to show that a given food substance contains protein.(b)Show how you will use(i)Modified Gabriel’s Synthsis(ii)Streckers’s Synthesis to prepare phenylalanine in the laboratory.arrow_forwardIllustrate a chemical equation showing the following:1. Hydrogenation of triarachidonin to Triarachidin in the presence of Nickel2. Hydrolysis of alpha-stearo-beta-oleoarachidonate3. Hydrolysis of tricaprin4. Saponification of tripalmitin with 3 moles NaOHarrow_forwardThe final steps in the Edman degradation result in rearrangement of a thiazolinone to an N-phenylthiohydantoin.Draw a stepwise mechanism for this acid-catalyzed reaction.arrow_forward
- Carboxypeptidase requires a Zn²+ cofactor for the hydrolysis of the peptide bond of a C-terminal aromatic amino acid. T/F?arrow_forwardWhat would be the structure of the two anomers of idopyranose in HAWORTH FORMULA? a. its beta-anomer and b. its alpha-anomerarrow_forwardIllustrate the treatment of methyl α-D-glucopyranoside with aqueous acid forms a mixture of α- and β-D-glucose and methanol ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you


Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY