
Concept explainers
(a)
Interpretation:
The product formed by the reaction of p-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various
Answer to Problem 25.61P
The product formed by the reaction of p-methylaniline
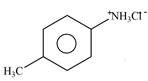
Explanation of Solution
The given reagent is
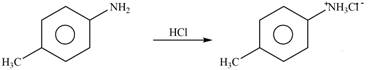
Figure 1
The nitrogen atom of p-methylaniline is basic in nature. It abstracts proton from
The product formed by the reaction of p-methylaniline
(b)
Interpretation:
The product formed by the reaction of p-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of organic chemistry. It can be a simple one-step reaction or it may involve many steps.
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various chemical reactions like reduction, oxidation, base, catalysis, and the addition of functional groups.
Answer to Problem 25.61P
The products formed by the reaction of p-methylaniline
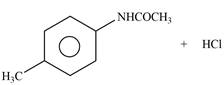
Explanation of Solution
The given reagent is
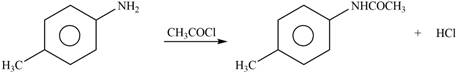
Figure 2
In the above reaction, acetyl chloride dissociates to form
The products formed by the reaction of p-methylaniline
(c)
Interpretation:
The product formed by the reaction of p-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of organic chemistry. It can be a simple one-step reaction or it may involve many steps.
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various chemical reactions like reduction, oxidation, base, catalysis, and the addition of functional groups.
Answer to Problem 25.61P
The products formed by the reaction of p-methylaniline
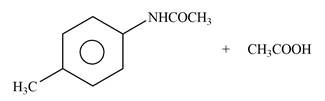
Explanation of Solution
The given reagent is
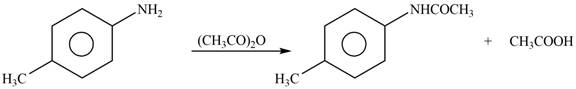
Figure 3
In the above reaction, the nucleophilic nitrogen atom attacks at the electrophilic carbonyl carbon atom of acetic anhydride to form the final product amide.
The products formed by the reaction of p-methylaniline
(d)
Interpretation:
The product formed by the reaction of p-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of organic chemistry. It can be a simple one-step reaction or it may involve many steps.
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various chemical reactions like reduction, oxidation, base, catalysis, and the addition of functional groups.
Answer to Problem 25.61P
The product formed by the reaction of p-methylaniline
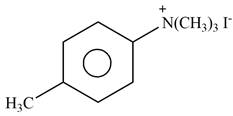
Explanation of Solution
The given reagent is excess
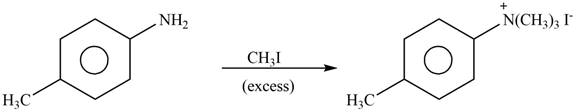
Figure 4
In the above reaction, p-methylaniline recats with excess of methyl iodide to form
The product formed by the reaction of p-methylaniline
(e)
Interpretation:
The product formed by the reaction of p-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of organic chemistry. It can be a simple one-step reaction or it may involve many steps.
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various chemical reactions like reduction, oxidation, base, catalysis, and the addition of functional groups.
Answer to Problem 25.61P
The products formed by the reaction of p-methylaniline
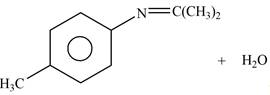
Explanation of Solution
The given reagent is
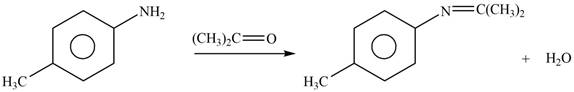
Figure 5
In the above reaction, p-methylaniline reacts with acetone to form the final product.
The products formed by the reaction of p-methylaniline
(f)
Interpretation:
The product formed by the reaction of p-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of organic chemistry. It can be a simple one-step reaction or it may involve many steps.
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various chemical reactions like reduction, oxidation, base, catalysis, and the addition of functional groups.
Answer to Problem 25.61P
The product formed by the reaction of p-methylaniline
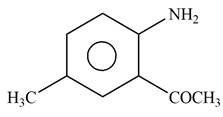
Explanation of Solution
The given reagent is
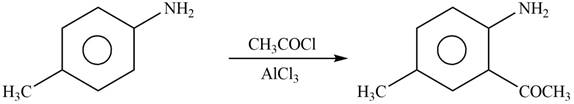
Figure 6
In the above reaction, acetyl chloride dissociates to form
The product formed by the reaction of p-methylaniline
(g)
Interpretation:
The product formed by the reaction of p-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of organic chemistry. It can be a simple one-step reaction or it may involve many steps.
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various chemical reactions like reduction, oxidation, base, catalysis, and the addition of functional groups.
Answer to Problem 25.61P
The product formed by the reaction of p-methylaniline
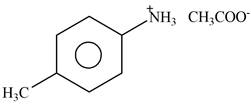
Explanation of Solution
The given reagent is
Figure 7
The nitrogen atom of p-methylaniline is basic in nature. It abstracts proton from
The product formed by the reaction of p-methylaniline
(h)
Interpretation:
The product formed by the reaction of p-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of organic chemistry. It can be a simple one-step reaction or it may involve many steps.
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various chemical reactions like reduction, oxidation, base, catalysis, and the addition of functional groups.
Answer to Problem 25.61P
The product formed by the reaction of p-methylaniline
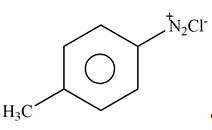
Explanation of Solution
The given reagent is
Figure 8
Diazonium salts are prepared by the treatment of
: The product formed by the reaction of p-methylaniline
(i)
Interpretation:
The product formed by the reaction of p-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of organic chemistry. It can be a simple one-step reaction or it may involve many steps.
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various chemical reactions like reduction, oxidation, base, catalysis, and the addition of functional groups.
Answer to Problem 25.61P
The product formed by the reaction of p-methylaniline
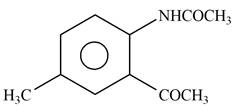
Explanation of Solution
The given reagent is
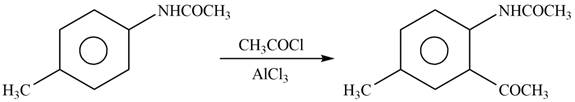
Figure 9
In the above reaction, acetyl chloride dissociates to form
The product formed by the reaction of p-methylaniline
(j)
Interpretation:
The product formed by the reaction of P-methylaniline
Concept introduction:
Synthesis is one of the major areas in the field of organic chemistry. It can be a simple one-step reaction or it may involve many steps.
The inorganic reagents do not contain a carbon atom. These reagents generally contain inorganic molecules, but sometimes small organic molecule also acts as an inorganic reagent. The reagents are additional chemical substances that are used to perform various chemical reactions like reduction, oxidation, base, catalysis, and the addition of functional groups.
Answer to Problem 25.61P
The product formed by the reaction of p-methylaniline
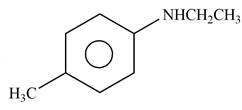
Explanation of Solution
The given reagent is
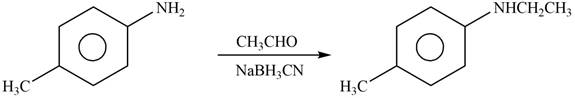
Figure 10
When an
The product formed by the reaction of p-methylaniline
Want to see more full solutions like this?
Chapter 25 Solutions
Organic Chemistry -Study Guide / Solution Manual (Custom)
- What is the correct reagent?arrow_forwardWhich reagents are used for reaction 1? a NaN3, ethanol and NaBH4, ethanol, H3O+ b CH3CH2NH2, NaOH c NH3, NaOH d NH3, DCC Which reagent are used for reaction 2? a CH3COOH, DCC b CH3COCH3 and NaBH4, ethanol, H3O+ c CH3COH and NaBH4, ethanol, H3O+ d CH3CONH2 and CH3CH2NH2arrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forward
- 3. a. What is the chemical structure of benzoic acid, circle functional groups different than alkane,alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? __________________________arrow_forwardDraw the major product of the reaction between 1-butanol and Na2Cr2O7, H2SO4, H2O.arrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. a. HNO3, H2SO4 h. product in (a), then Sn, HClarrow_forward
- 1. O-hydroxybenzoic acid is a major product formed with phenol and which other reactant/s I-primary alcohol II-sodium hydroxide III-water IV-carbon dioxide A.I and III B. I and IV C. II and III D. II and IVarrow_forwardDraw the structure of the predominant form of CF3CH2OH (pK a = 12.4) at pH = 6.arrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OHarrow_forward
- Draw the missing starting material. Reagent 1 is benzene and AlCl3. Reagent B is Zn(Hg) and HCl.arrow_forward(C₈H₁₀) according to the following reaction: C₈H₁₀(l) + O₂(g) + NH₃(g) → C₈H₄N₂(s) + H₂O(l) How many grams of water would be produced by the complete ammoxidation of 16.50 moles of o-xylene?arrow_forwardWhat reagent is necessary to complete the reaction? CH3-CH2-CH-C-OH ? CHỊCH,CH C-0- Na* CH3 CH3 NaO O NaCl O Na O NaOH + H₂Oarrow_forward
