
Synthesize each compound from benzene. Use a diazonium salt as one of the synthetic intermediates.
a. 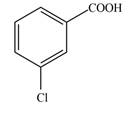 c.
c. 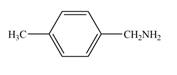 e.
e.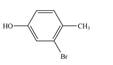
b. 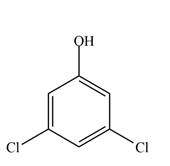 d.
d. 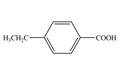 f.
f.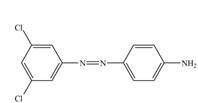
(a)
Interpretation: The given compound is to be synthesized from benzene by the use of diazonium salt as one of the synthetic intermediates.
Concept introduction: Amines are the derivatives of ammonia consisting of nitrogen atom with the lone pair of electrons. They are basic compounds. The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 25.80P
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
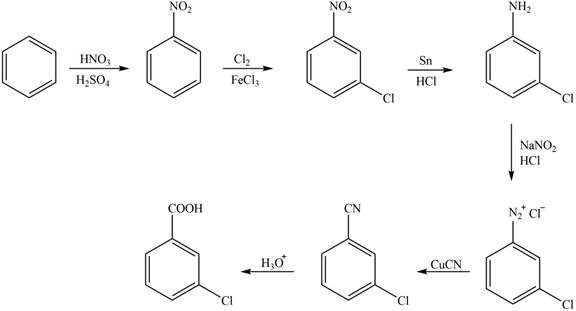
Explanation of Solution
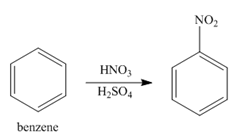
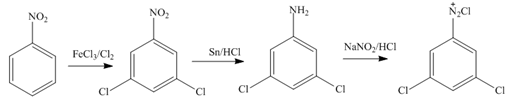
In the first step of the synthesis, nitration of benzene ring takes place. The nitro group is meta directing, as a result the chlorination takes place at meta position in the second step. The nitro group converts to amine group on reaction with
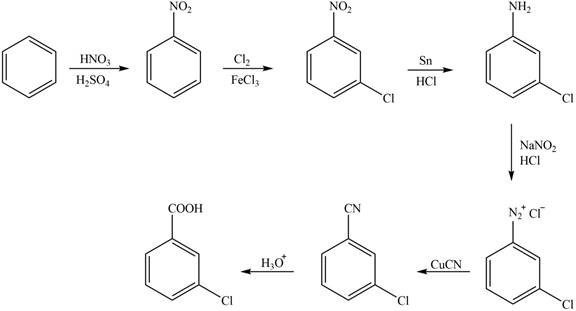
Figure 1
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown in Figure 1.
(b)
Interpretation: The given compound is to be synthesized from benzene by the use of diazonium salt as one of the synthetic intermediates.
Concept introduction: Amines are the derivatives of ammonia consisting of nitrogen atom with the lone pair of electrons. They are basic compounds. The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 25.80P
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
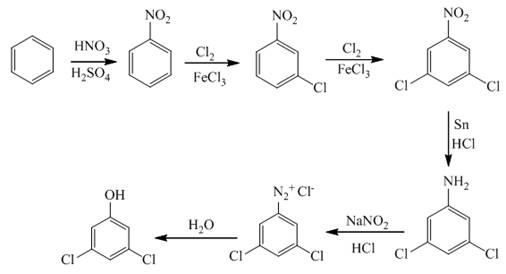
Explanation of Solution
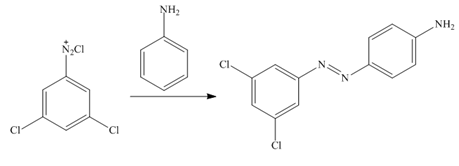
In the first step of the synthesis, nitration of benzene ring takes place. The nitro group is meta directing, as a result the chlorination takes place at meta position in the next two steps. The nitro group converts to amine group on reaction with
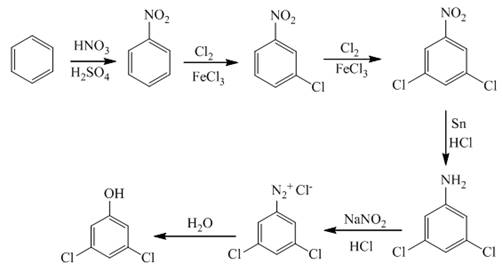
Figure 2
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown in Figure 2.
(c)
Interpretation: The given compound is to be synthesized from benzene by the use of diazonium salt as one of the synthetic intermediates.
Concept introduction: Amines are the derivatives of ammonia consisting of nitrogen atom with the lone pair of electrons. They are basic compounds. The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 25.80P
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
Explanation of Solution
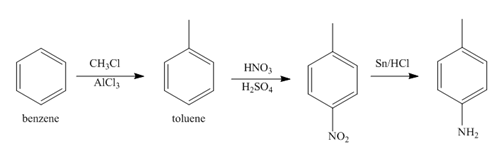
The first step of synthesis of given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
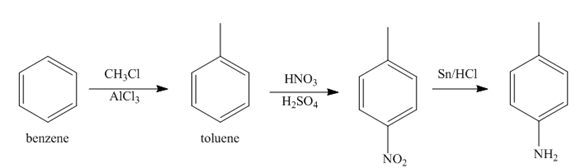
Figure 3
In the above step, benzene undergoes Friedel craft’s alkylation reaction to form toluene. It further reacts with
The second step of synthesis of given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
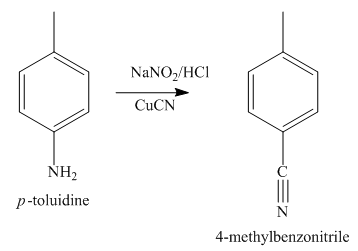
Figure 4
The compound
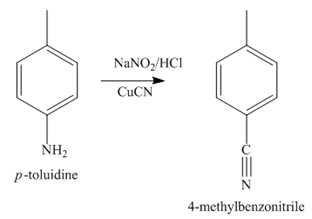
The third step of synthesis of given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
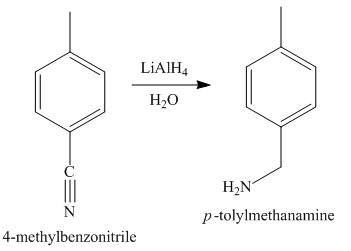
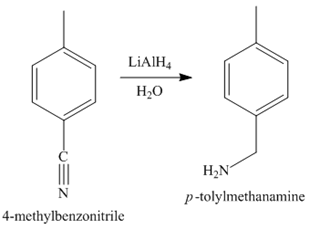
Figure 5
In the last step, the reduction of nitrile to amine takes place in the presence of
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown in Figure 3, Figure 4 and Figure 5.
(d)
Interpretation: The given compound is to be synthesized from benzene by the use of diazonium salt as one of the synthetic intermediates.
Concept introduction: Amines are the derivatives of ammonia consisting of a nitrogen atom with the lone pair of electrons. They are basic compounds. The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 25.80P
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
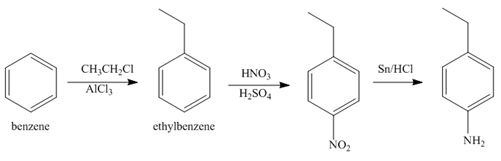
Explanation of Solution
The first step of synthesis of given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
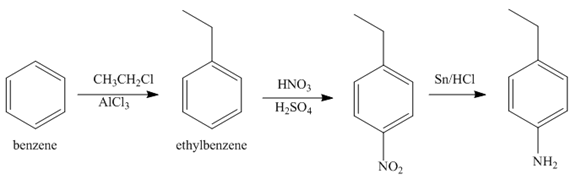
Figure 6
In the above step, benzene undergoes Friedel craft’s alkylation reaction to form ethylbenzene. It further reacts with
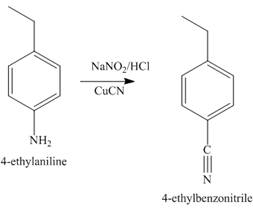
The second step of synthesis of given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
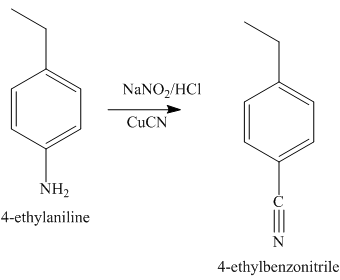
Figure 7
The compound
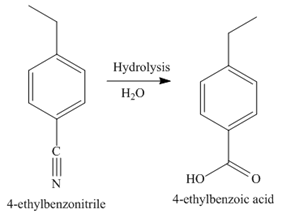
The third step of synthesis of given compound from benzene, by the use of a diazonium salt as one of the synthetic intermediates, is shown below.
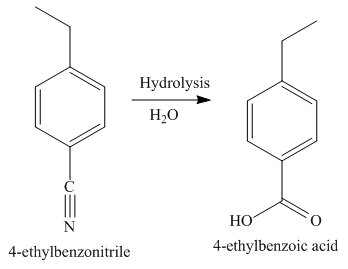
Figure 8
In the last step, the hydrolysis reaction takes place, which results in the formation of
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown in Figure 6, Figure 7 and Figure 8.
(e)
Interpretation: The given compound is to be synthesized from benzene by the use of diazonium salt as one of the synthetic intermediates.
Concept introduction: Amines are the derivatives of ammonia consisting of nitrogen atom with the lone pair of electrons. They are basic compounds. The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 25.80P
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
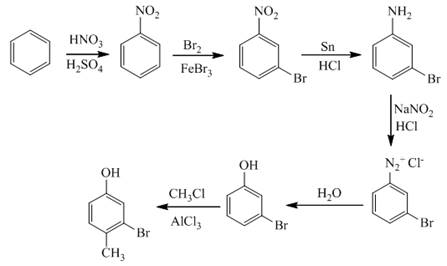
Explanation of Solution
In the first step of the synthesis, nitration of benzene ring takes place. The nitro group is meta directing, as a result the bromination takes place at meta position in the second step. The nitro group converts to amine group on reaction with
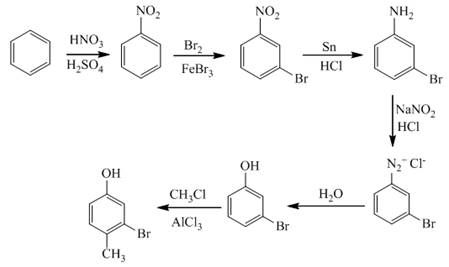
Figure 9
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown in Figure 9.
(f)
Interpretation: The given compound is to be synthesized from benzene by the use of diazonium salt as one of the synthetic intermediates.
Concept introduction: Amines are the derivatives of ammonia consisting of a nitrogen atom with the lone pair of electrons. They are basic compounds. The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 25.80P
The synthesis of given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates is shown below.
Explanation of Solution
The first step of synthesis of given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
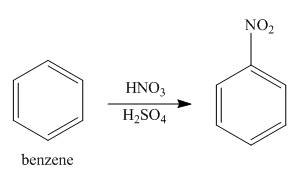
Figure 10
In the above step, benzene reacts with
The second step of synthesis of given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
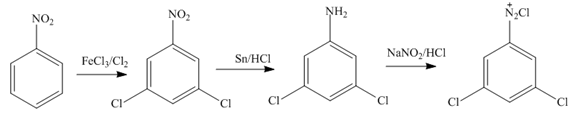
Figure 11
Nitrobenzene undergoes chlorination and reduction reaction to form
The third step of synthesis of given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown below.
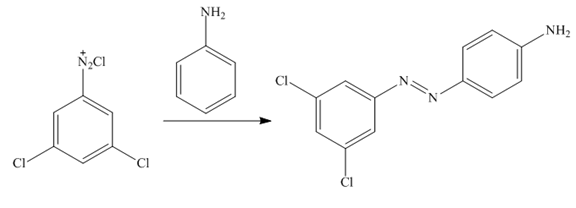
Figure 12
In the last step,
The synthesis of a given compound from benzene, by the use of diazonium salt as one of the synthetic intermediates, is shown in Figure 10, Figure 11 and Figure 12.
Want to see more full solutions like this?
Chapter 25 Solutions
Organic Chemistry -Study Guide / Solution Manual (Custom)
- A key step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass found in tropical and subtropical regions of the world, involved the reaction of compound A and dihalide B with two equivalents of LDA to form C. Draw a stepwise mechanism for this reaction. β-Vetivone contains a spiro ring system—that is, two rings that share a single carbon atom.arrow_forwardDraw a stepwise mechanism for the attached reaction, which involves two Friedel–Crafts reactions. B was an intermediate in the synthesis of the antidepressant sertralinearrow_forwardDevise a synthesis of attached alkyne. You may use acetylene, benzene,organic halides, ethylene oxide, and any other required inorganicreagents.arrow_forward
- What product is formed when A is treated with Grubbs catalyst underhigh-dilution conditions? This reaction was a key step in the synthesis ofstemoamide, the naturally occurring amide described in the opening paragraph.arrow_forwardShow how you might use the Williamson ether synthesis to prepare each ether. (a) (b)arrow_forwardNot all aldehyde give a positve Bendicts test. Which of the follwing aldehydes do? a. d. b. e. c.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


