
Concept explainers
In addition to organic halides, alkyl tosylates (
a. 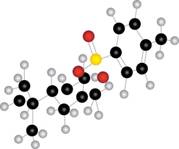 b.
b. 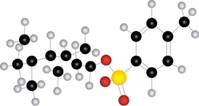
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
ORGANIC CHEMISTRY
- When (ft)-6-bromo-2,6-dimethylnonane is dissolved in CH3OH, nucleophilic substitution yields an optically inactive solution. When the isomeric halide (fl)-2-bromo-2,5- dimethylnonane is dissolved in CH3OH under the same conditions, nucleophilic substitution forms an optically active solution. Draw the products formed in each reaction, and explain why the difference in optical activity is observed.arrow_forwardCompound A is first reacted with methylamine in the presence of acid and then treated with NaBH3CN. Using the spectroscopic data given, what is the structure of the product after step 1?arrow_forwardIdentify compounds A – E of the reaction sequence shown in Scheme III, making sure to include stereochemistry as appropriate.a) Identify compound A in Scheme III. b) Identify compound B in Scheme III. c) Identify compound C in Scheme III. d) Identify compound D in Scheme III. e) Identify compound E in Scheme III.arrow_forward
- This is not A or C. The second one is not A or Darrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. CH3COCl, AlCl3arrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. Part (b), then CH3COCl, AlCl3arrow_forward
- Please answer BOTH questions, thanks so much!arrow_forwardBe sure to answer all parts. What is the major product formed when the following alkyl halide is treated with each of the following reagents: [1] NaOCOCH3; [2] NaOCH3; [3] KOC(CH3)3? If it is not possible to predict the major product, identify the products in the mixture and the mechanism by which each is formed. Part 1: It is possible to predict the major product for: NaOCOCH3 NaOCH3 KOC(CH3)3 none of the reagents Part 2 out of 2 Major product with [1] NaOCOCH3: Major product with [2] NaOCH3: Major product with [3] KOC(CH3)3: Check my work edit structure ... draw structure ... draw structure ... Next part Hint ?arrow_forwardAnswer the following questions about curcumin, a yellow pigment isolated from turmeric, a tropical perennial in the ginger family and a principal ingredient in curry powder.a.In Chapter 11, we learned that most enols, compounds that contain a hydroxy group bonded to a C=C, are unstable and tautomerize to carbonyl groups. Draw the keto form of the enol of curcumin, and explain why the enol is more stable than many other enols. b.Explain why the enol O—H proton is more acidic than an alcohol O—H proton. c. Why is curcumin colored? d.Explain why curcumin is an antioxidant.arrow_forward
- Part A Provide the reagents necessary to complete the following transformation. CH2CH3 CH,CH3 enantiomer O H2O, H2SO4 O mСРВА О 1) ВНз * ТHF 2) H2O2, Но- Os04, H2O2 О 1) СН3СОЗ Н 2) Н*, Н2Оarrow_forwardDraw all products, including stereoisomers, in attached reaction.arrow_forwardThe product below can be prepared from cyclohexanone through the following reaction sequence? A) [1] (CH3CH2)2NH, mild acid; (2) CH3CH2CH2BR; (3] HCI, H20 B) [1] CH3CH2NH2, mild acid; [2] CH3CH2CH2Br; [3] HCI, H20 ) [1] CH3OH, H+; [2] CH3CH2CH2MgBr; [3] HCI, H20 D) [1] nBuLi; (2] CH3CH2CH2Br: (3] ETNH2, mild acid E) [1] nBuLi: (2] CH3CH2CH2Br; (3] Et2NH, mild acidarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


