
Concept explainers
Interpretation:
All resonance structures for the product of head-to-tail addition shown in Figure 26-11 are to be drawn. Then curved-arrow notation is to be used to show the movement of electrons in the head-to-tail addition of a growing chain of poly(ethyl acrylate) to a molecule of ethyl acrylate. The resonance structures are to be drawn to explain why head-to tail addition occurs instead of head-to-head addition.
Concept introduction:
Styrene is an unsymmetric vinyl monomer, so we can differentiate between the two carbons of the vinyl group. The less substituted carbon is called the tail, and the more substituted carbon is called the head. As a result, regiochemistry comes into play during
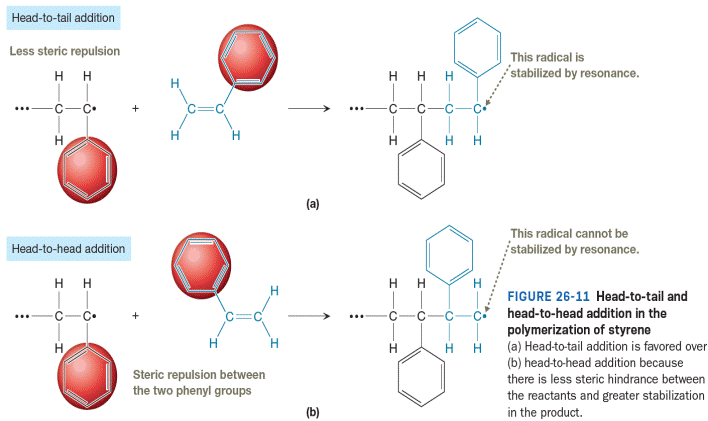
The head-to-tail addition is favored in the polymerization of polystyrene for the two reasons shown in Figure 26-11. First, steric repulsion between the two phenyl groups (one at the end of the propagating radical and one in the monomer) makes it less likely that a head-to-head collision will bring the bond-forming carbons within a bond length. Secondly, the radical formed in the head-to-tail addition is more stable. The radical produced from head-to-tail addition is resonance stabilized while the radical produced from head-to-head addition is not.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- Based on IR spectra of "Free Radical Polymerization of Styrene" experiment, how well the reaction workedarrow_forwardWhy do you think initiator molecule is needed to induce the polymerization of ethylenearrow_forwardShow the structure of the polymer that results from heating the following diepoxide and diamine:arrow_forward
- what chemical reactions should not be performed on Merrifield polystyrene resin?arrow_forwardShow the intermediate that would result if the growing chain added to the other end of the styrene double bond. Explain why the final polymer has phenyl groups substituted on every other carbon atom rather than randomly distributedarrow_forwardwhat is electrical double layer ? how it is formed ? how many types of micelles ate there give their application. asap fast or else skiparrow_forward
- 1.Discuss the suitable initiator to start an addition polymerization. 2.Define what is auto-acceleration, what causes this effect and how to minimize this effect. 3.Briefly discuss emulsion and suspension polymerization techniques.arrow_forwardThis is correct for the synthesis of Nylon 8,10?arrow_forwardProvide missing information: The IUPAC name of the structure below is ........................... This structure is a glycol (input TRUE or FALSE)....................arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

