
Concept explainers
Interpretation:
The structure of triphosphate of guanosine has to be drawn.
Concept Introduction:
Composition of
Sugar: In both DNA and RNA, sugar portion is found. In DNA, the sugar is D-ribose, where at 2’hydroxyl group is absent and in RNA, the hydroxyl group is present at 2’.
Nitrogenous bases: Five types of nitrogenous bases (has unique one-letter code A, G, T, U, and C) are derived from two parent compounds called purine and pyrimidine. The purine derivatives are Adenine and Guanine are two fused nitrogen containing rings. The pyrimidine derivatives are Thymine, Cytosine, and Uracil are only one nitrogen containing six-membered ring. Adenine, Guanine, Thymine, and Cytosine are the nitrogenous bases present in DNA. Adenine, Guanine, Cytosine and Uracil are the nitrogenous bases present in RNA.
Nucleotide: (Nucleoside + phosphate)
Nucleotides are the building blocks of nuclei acids; monomers of DNA and RNA
Nucleoside and its naming: The combination of monosaccharide (sugar) and nitrogenous base is known as nucleoside. The nucleoside names are the nitrogenous base name modified with criteria. While naming nucleoside of purine derivatives the suffix ‘-osine’ is included and for pyrimidine derivatives the suffix ‘-idine’ is used. No prefix used for the nucleosides containing ribose and the prefix ‘deoxy-’ is used for deoxyribose.
Naming nucleotide: At the end of the nucleoside, phosphate group is added. For example, 5’-monophosphate means adding one phosphate group at 5’carbon in the sugar ring. Uridine monophosphate can be written as UMP.
Numbering the atoms in sugar and base rings:
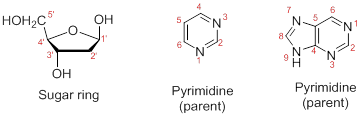
In order to distinguish the atoms in the sugar of a nucleoside and atoms of a base ring, numbers without prime is used for atoms in the base ring and numbers with prime used for the atoms in the sugar ring.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
EP FUND.OF GENERAL,ORG...-MOD.MASTERING
- What is the significance of substrate-level phosphorylation reactions? Which of the reactions in the citric acid cycle involve a substrate-level phosphorylation? Name another example from a biochemical pathway with which you are familiar.arrow_forwardMyristoleic acid is a monounsaturated fatty acid found in small amounts in a variety of foods. Calculate the net ATP yield from the complete β-oxidation of myristoleic acid. The formula of myristoleic acid is shown below (it is assumed that the total ATP production is the same for both saturated and unsaturated fatty acids having the same carbon chain length). CH3-(CH2)3-CHCH-(CH2)7-COOH (Given: The oxidation of one NADH yields 2.5 ATP; the oxidation of one FADH2 yields 1.5 ATP; and the oxidation of one acetyl CoA yields 10 ATP. ) Group of answer choices a. 96 ATP b. 92 ATP c. 94 ATP d. 34 ATP e. 36 ATParrow_forwardName the molecules that conserve most of the energy from the redox reactions of the citric acid cycle (see Figure 9.12). How is this energy converted to a form that can be used to make ATP?arrow_forward
- List four different products of citric acid cycle.arrow_forwardThe glycerol 3-phosphate required for the synthesis of glycerophospholipids can be synthesized from a glycolytic intermediate. Propose a reaction sequence for this conversionarrow_forwardWhat substance acts as the starting point of the citric acid cycle, reacting with acetyl-CoA in the first step and being regenerated in the last step? Draw its structure.arrow_forward
- Decylic acid is a saturated fatty acid that occurs naturally in coconut oil and palm kernel oil. Calculate the net ATP yield when decylic acid undergoes complete B oxidation. The formula of decylic acid is shown below: (Given: The oxidation of one NADH yields 2.5 ATP; the oxidation of one FADH2 yields 1.5 ATP; and the oxidation of one acetyl CoA yields 10 ATP.) O 50 ATP O 52 ATP 66 ATP OH O 64 ATParrow_forwardCells often use the same enzyme reaction pattern for analogous metabolic conversions. For example, the steps in the oxidation of pyruvate to acetyl-CoA and of a-ketoglutarate to succinyl-CoA, although catalyzed by different enzymes, are very similar. The first stage of fatty acid oxidation follows a reaction sequence closely resembling a sequence in the citric acid cycle. Use equations to show the analogous reaction sequences in the two pathways.arrow_forwardFumarate produced in step 6 of the citric acid cycle must have a trans double bond to continue on in the cycle. Suggest a reason why the corresponding cis double-bond isomer cannot continue in the cycle.arrow_forward
- Identify at least five biological effects of disorders of the citric acid cyclearrow_forward(a) Convert the ball-and-stick model of B, one of the eight synthetic intermediates in the citric acid cycle, to a structural formula, and name the compound. (b) What is the immediate precursor of B in the citric acid cycle; that is, what compound forms B as a reaction product? (c) What compound is formed from B in the citric acid cycle?arrow_forwardPhosphoglycerate mutase transfers a phosphoryl group from C3 of 3-phosphoglycerate to C2 position to give 2-phosphoglycerate. Why is it important to have the phosphoryl group at position 2 rather than at C3? It was observed that the activity of some preparations of the enzyme could be increased if catalytic (very small) amount of 2, 3-bisphosphoglycerate was added to the enzyme. Give a possible explanation for this observation.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





