
(a)
Interpretation:
Each of the following reactions involves a sequence of two pericyclic reactions. The intermediate X involved in each reaction, and pericyclic reactions involved are to be depicted.
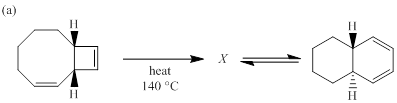
Concept introduction:
Electrocyclic reactions are a pericyclic reaction which occur intramolecularly. These reactions will result in the formation of ring compounds under the influence of heat or light. Notably, in this process one new sigma bond is formed and one old π-bond is consumed. Intriguingly, the reverse ring opening electrocyclic reaction can also be possible to occur under the same reaction mechanism but in reverse manner. In phase orbital overlap results in symmetry allowed electrocyclic reactions. Selection rules of electrocyclic reactions are;
| No. of electrons | Activation mode | Stereochemistry of rotation |
| 4n | Thermal Photochemical |
Con Dis |
| 4n + 2 | Thermal Photochemical |
Dis Con |
(b)
Interpretation:
Each of the following reactions involves a sequence of two pericyclic reactions. The intermediate Y involved in each reaction, and pericyclic reactions involved are to be depicted.
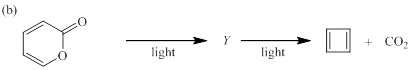
Concept introduction:
Electrocyclic reactions are a pericyclic reaction which occur intramolecularly. These reactions will result in the formation of ring compounds under the influence of heat or light. Notably, in this process one new sigma bond is formed and one old π-bond is consumed. Intriguingly, the reverse ring opening electrocyclic reaction can also be possible to occur under the same reaction mechanism but in reverse manner.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
EBK ORGANIC CHEMISTRY
- A problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate: Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:arrow_forwardSynthesize the product from the given material. Give the reagents necessary and draw out any intermediate products along the way.arrow_forwardSynthesize the products by drawing out reagents and intermediates along the way.arrow_forward
- The transformation takes place via two sequential pericyclic reactions. Identify the two reactions and give a critical explanation whether the reactions are allowable or not. Explain the stereochemistry.arrow_forwardWhen cyclohexene is reacted with hydrobromic acid in acetic acid, the major product is bromocyclohexane. There is a small amount of cyclohexyl acetate formed. What is the mechanism that forms both compounds? What is the purification procedure that isolates both compounds?arrow_forwardIndicate the reagents and conditions to carry out the following transformations.arrow_forward
- Heterocyclic compounds can undergo nitration reactions with some regioselectivity, giving products in majority. From the following reaction give the raw material from which it is formed and the mechanism under which the transformation takes place.arrow_forwardWhich of the following statements is FALSE about pericyclic reactions? * Pericyclic reactions occur in a single step. The transition state of pericyclic reactions is cyclic. The outcome of pericyclic reactions vary depending on the identity of the solvent used. O There are no intermediates in a pericyclic reaction.arrow_forwardBr Brz CH3 CH3 H3C CH2CI2 H3C Br Electrophilic addition of bromine, Br2; to alkenes yields a 1,2-dibromoalkane. The reaction proceeds through a cyclic intermediate known as a bromonium ion. The reaction occurs in an anhydrous solvent such as CH,Cl). In the second step of the reaction, bromide is the nucleophile and attacks at one of the carbons of the bromonium ion to yield the product. Due to steric clashes, the bromide ion always attacks the carbon from the opposite face of the bromonium ion so that a product with anti stereochemistry is formed. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Br: :Br: .CH3 H3C H3C CH3 Br:arrow_forward
