
Concept explainers
(a)
Interpretation: The number of MOs in the given compound
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
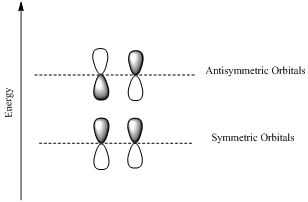
(b)
Interpretation: The designation of HOMO for the given molecule’s molecular orbital has to be given.
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
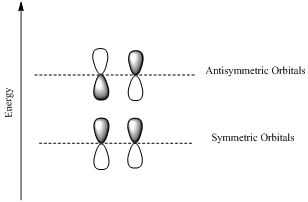
(c)
Interpretation: Number of nodes in the given molecule has to be given.
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
Node is the site with zero electron density.
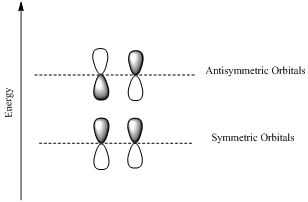
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
ORGANIC CHEMISTRY W/ST.GDE W/ACCESS
- Consider any one of the four identical hybrid orbitals in the 109.5° set. a. What fraction of the clay in this hybrid orbital was originally red (carne from the 2s orbital)? b. What fraction of the clay in this hybrid orbital was originally green (carne from a 2porbital)? c. Explain the name “ s(1/4)p(3/4) hybrid orbital” for each of the four orbitals. d. In fact, each of the four hybrid orbitals in the 109.5° set is called an sp3 -hybrid orbital.Explain the name “ sp3 -hybrid orbital.”arrow_forwardDraw one all-bonding MO, thena pair of degenerate MOs, and then a final pair of degenerate MOs. Draw the energydiagram, fill in the electrons, and confirm the electronic configurations of the cyclopentadienyl cation and anionarrow_forwardAnswer the following questions for the MOs of 1,3-butadiene: a. Which are p bonding MOs, and which are p* antibonding MOs? b. Which MOs are symmetric, and which are antisymmetric? c. Which MO is the HOMO and which is the LUMO in the ground state? d. Which MO is the HOMO and which is the LUMO in the excited state? e. What is the relationship between the HOMO and the LUMO and symmetric and antisymmetric orbitals?arrow_forward
- The pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energy. (continued)762 CHAPTER 15 Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy(e) Show how many electrons are in each MO for the pentadienyl radical (ground state).(f) Show how your molecular orbital picture agrees with the resonance picture showing delocalization of the unpairedelectron onto three carbon atoms.(g) Remove the highest-energy electron from the pentadienyl radical to give the pentadienyl cation. Which carbon atomsshare the positive charge? Does this picture agree with the resonance picture?(h) Add an…arrow_forwardThe pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energyarrow_forwardHow many equivalent hydrogens are in CH2BrCl? (i.e., do they have identical environments with respect to the other atoms adjacent to themselves). A) 0 B) 3 C) 1 D) 2arrow_forward
- How would you develop a molecular orbital for a structure like buta-2-diene that has an anion on the first carbon and a cation on the fourth carbon? Also, how would MO look like for 2-dimethyl-1,3-pentadiene?arrow_forwardAnswer the following questions for 1,3,5- hexatriene, the conjugated triene containing six carbons.Which MOs are bonding? Which are antibonding?arrow_forwardAssign the stereochemical configuration (R or S) for each molecule. Show your work, indicating clearly which groups are assigned which priorities (#1 through #4) (e.g. by including the priority # directly adjacent to one of the atoms in that group).arrow_forward
- 1. Consider the following eqm: SO2(g) + NO2(g) ⇌ NO(g) + SO3(g)What effect will adding SO3 have on the rxn?What effect will removing NO2 have on the rxn? 2. Consider the following exothermic rxn at eqm: C3H8(g) + 5O2(g) ⇌ 4H2O(l) + 3CO2(g)What effect will increasing the temperature have on the rxn?What effect will increasing the pressure have on the rxn?arrow_forwardUse the experimental evidence to make areduction potential table for the 4 couples involved.(This one is more challenging because you have to figure out the couples yourself).V3+ + PuO2+ VO2+ + Pu4+Mo3+ + V3+ no reactionGa + Mo3+ Mo + Ga3+arrow_forwardPropanal (bp 48°C) and propanol (bp 97°C), both found on Table 4.2, have very similar surfaceareas and dipole moments. Construct an explanation for the large difference in boiling pointsbetween the two.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
