
Pushing Electrons
4th Edition
ISBN: 9781133951889
Author: Weeks, Daniel P.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3, Problem 14EQ
Interpretation Introduction
Interpretation:
The leaving group in the given molecule is to be determined in order identify the positively charged and neutral fragments produced during mechanism.
Concept Introduction:
Heterolytic cleavage of sigma bond occurs under a variety of conditions. The mechanism of bond cleavage is as shown below,
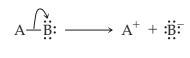
The arrow in the above mechanism indicates that the sigma electrons form A-B bond are leaving A and becoming the exclusive property of B. Since the fragment A is formally losing one electron, it must become positively charged and B must become negatively charged since it gains an electron.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Chemistry
For the following molecules, determine whether the double bond is E, Z, or neither.
Without consulting tables of data, on the same graph sketch the effective potential energy curves for H2 and He+2.
Qualitatively, how will the potential energy diagram for H2 be altered for N2? Draw one qualitative figure showing the potential energy curve for H2, and then add another curve for N2. Briefly explain how and why they differ.
Chapter 3 Solutions
Pushing Electrons
Ch. 3 - Prob. 1EQCh. 3 - Prob. 2EQCh. 3 - Prob. 3EQCh. 3 - Prob. 4EQCh. 3 - Prob. 5EQCh. 3 - Prob. 6EQCh. 3 - Here are some exercises in sigma bond breaking....Ch. 3 - Prob. 8EQCh. 3 - Prob. 9EQCh. 3 - Prob. 10EQ
Ch. 3 - Prob. 11EQCh. 3 - Prob. 12EQCh. 3 - Prob. 13EQCh. 3 - Prob. 14EQCh. 3 - Prob. 15EQCh. 3 - Prob. 16EQCh. 3 - Prob. 17EQCh. 3 - Prob. 18EQCh. 3 - Prob. 19EQCh. 3 - Prob. 20EQCh. 3 - Prob. 21EQCh. 3 - Prob. 22EQCh. 3 - Prob. 23EQCh. 3 - Prob. 24EQCh. 3 - Prob. 25EQCh. 3 - Prob. 26EQCh. 3 - Prob. 27EQCh. 3 - Prob. 28EQCh. 3 - Prob. 29EQCh. 3 - Prob. 30EQCh. 3 - The reaction just described is reversible....Ch. 3 - Prob. 32EQCh. 3 - Prob. 59EQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Use average bond enthalpies (linked above) to calculate the enthalpy change for the following gas-phase reaction. C2H4(g) + H2O2(g) --> CH2OHCH2OH(g) ΔHreaction = ?arrow_forwardThe force constant of a carbon monoxide bond is 1903 Nm-1, while the force constant of the bond in O2 is only 1176 Nm-1. Explain why the force constant of the CO bond is so much larger.arrow_forwardUse the bond energies provided to estimate DeltaH^ rxn for the reaction below. 2Br_(2(1))+C_(2)H_(2(g))rarrC_(2)H_(2)Br_(4(1))DeltaH^rxxn=?arrow_forward
- Which statement correctly describes the formation of the halogen radical, in radical halogenation? Options: Light excites electrons from the X2 lone pair into the anti-bonding orbital, thereby breaking the bond and forming radicals. Light excites electrons from the X2 sigma bond into the anti-bonding orbital, thereby breaking the bond and forming radicals. Heat excites electrons from the X2 lone pair into the anti-bonding orbital, thereby breaking the bond and forming radicals. Heat excites electrons from the X2 lone pair into the anti-bonding orbital, thereby breaking the bond and forming radicals.arrow_forwardIf the bond in the AB molecule is considered 100% ionic, the absolute value of the partial charge would be equal to the electron charge (1.602 x 10-19 C). If the distance between A and B atoms is taken as 10-10 m (1Å), the dipole moment between two elementary charges of opposite sign at atomic magnitude (1Å) distance between them is = 1.602x10-19 C x 10-10 m = 1.602 x 10- It is available in 29 Cm (or 4.8 x 10-18 esbcm). Dipole moment is given by debye (D) and 1D = 3.34x10-30 Cm (or 1D = 10-18 esbcm) (esb = electrostatic unit). 1. As can be seen, when the HCl covalent molecule is considered ionic and the bond length is 1.27x10-10 m, calculate the dipole moment. The experimental dipole moment of an HCl polar option is = 1.03 D. Calculate the value character of the H-Cl bond according to the bulletin. How many% covalent is the bond, how many% is ionic? Specifyarrow_forwardPlease send me the question in 20 minutes it's very urgent plz 8. Draw the Lewis structures for two resonance forms of the following moleculesions. Show any formal charges and determine which structure is major, which is minor, or if they are equivalenz [HICCO] (acylium ion)arrow_forward
- A student says "For molecules containing two different atoms, the magnitude of the partial charges is related to the difference in EN of those two atoms." Is the data in Table 1 consistent with this statement? Provide your answer using one or more grammatically correct sentences and include specific examples from Table 1 in your response. Does the data for H2O and H2S in Table 3 support this statement? Provide your answer using one or more grammatically correct sentences and specifically address the data in Table 3.arrow_forwardExplain the following observations about two carbon-oxygen bonds in the methane (formate) anion, HCO2-. You may draw a Lewis electron-dot diagram (or diagrams) of methanoate ion as part of your explanations. i. The two carbon-oxygen bonds in the methanoate(formate) anion, HCO2-, have the same length. ii.the length of the carbon-oxygen bonds in the methanoate(formate) anion, HCO2-, is intermediate between the length of the carbon-oxygen bond in methanol and the length of the carbon-oxygen bond in methanal.arrow_forwardWhat are the resonance strucutes for this molecule?arrow_forward
- How can we identify whether the compounds are ionic or covalent for part 1?arrow_forwardFor which of the following pairs would you expect ∆o to be larger and why? [MnF6]4- and [ReF6]4-arrow_forwardA radical is an unpaired electron – see the carbon radical from the last question, for example. Radicals are highly unstable and reactive. In the structure to the right, all formal charges are zero. Fill in all the non-bonding electrons and identify if the species is a radical.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you


 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning



Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY