
Concept explainers
(a)
Interpretation:
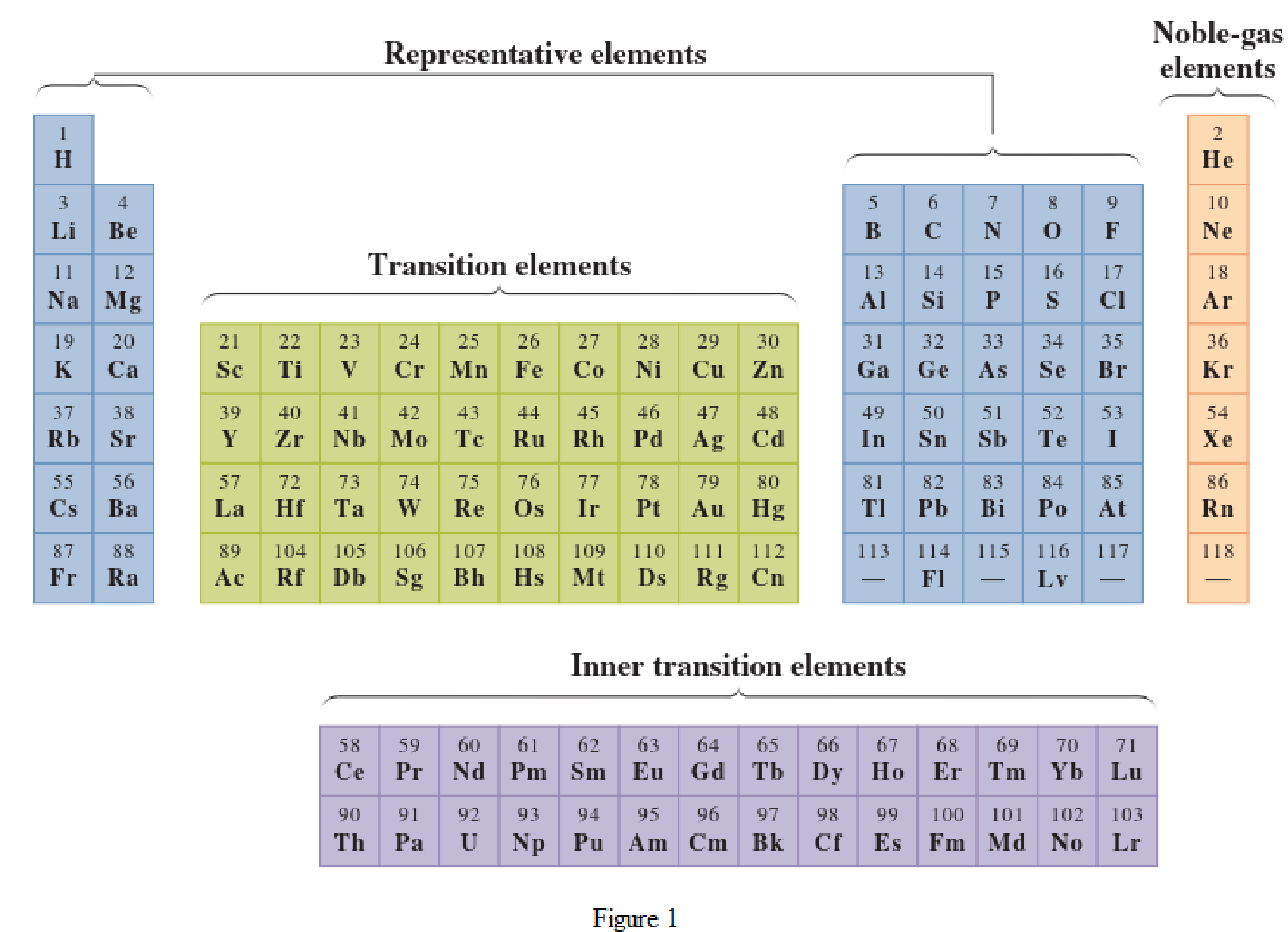
In the given periodic table, how many elements those are highlighted which represent representative elements have to be determined.
Concept Introduction:
Elements in the periodic table are classified in several different ways and out of them two most common systems are,
- System based on the physical properties in which they are classified as metals and nonmetals.
- System based on electronic configuration in which they are classified as noble-gas, representative elements,
transition elements , or inner-transition elements.
Noble-gas elements are the ones that are located in far right of periodic table. The physical state of these elements at room temperature is gas. The noble gases have their electronic configuration ending with
Representative elements are the ones that are in s area and area of the periodic table. They have partially filled s subshell or p subshell in their electronic configurations. Some of the elements are nonmetals while others are metals.
Transition elements are the ones that are located in d area of periodic table. They have the distinguishing electrons in their d subshell. All the transition elements are metals.
Inner transition elements are the ones that are located in f area of the periodic table. They have the distinguishing electrons in their f subshell. All inner transition elements are metals.
(b)
Interpretation:
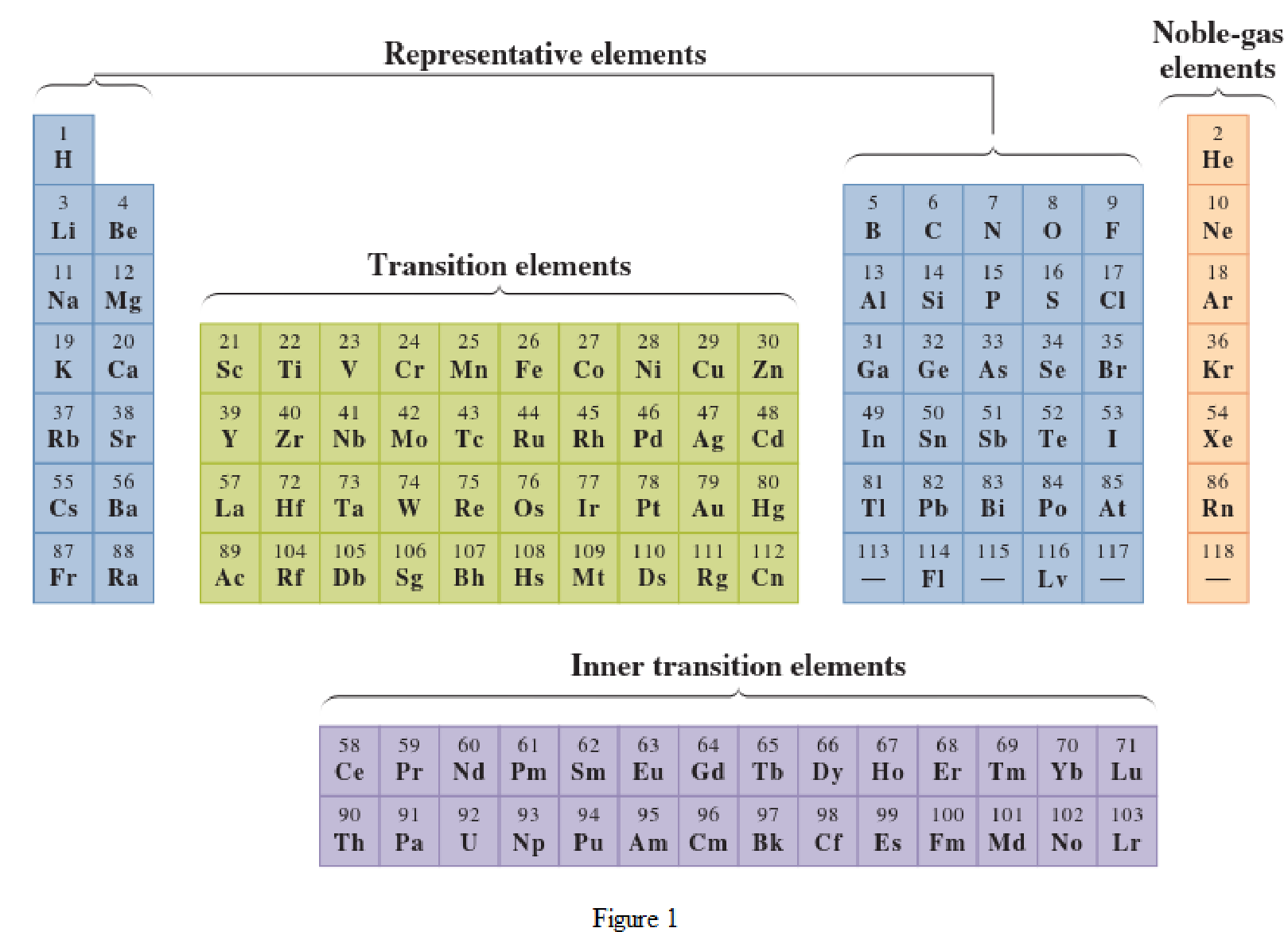
In the given periodic table, how many elements those are highlighted which represent noble gases has to be determined.
Concept Introduction:
Elements in the periodic table are classified in several different ways and out of them two most common systems are,
- System based on the physical properties in which they are classified as metals and nonmetals.
- System based on electronic configuration in which they are classified as noble-gas, representative elements, transition elements, or inner-transition elements.
Noble-gas elements are the ones that are located in far right of periodic table. The physical state of these elements at room temperature is gas. The noble gases have their electronic configuration ending with
Representative elements are the ones that are in s area and area of the periodic table. They have partially filled s subshell or p subshell in their electronic configurations. Some of the elements are nonmetals while others are metals.
Transition elements are the ones that are located in d area of periodic table. They have the distinguishing electrons in their d subshell. All the transition elements are metals.
Inner transition elements are the ones that are located in f area of the periodic table. They have the distinguishing electrons in their f subshell. All inner transition elements are metals.
(c)
Interpretation:
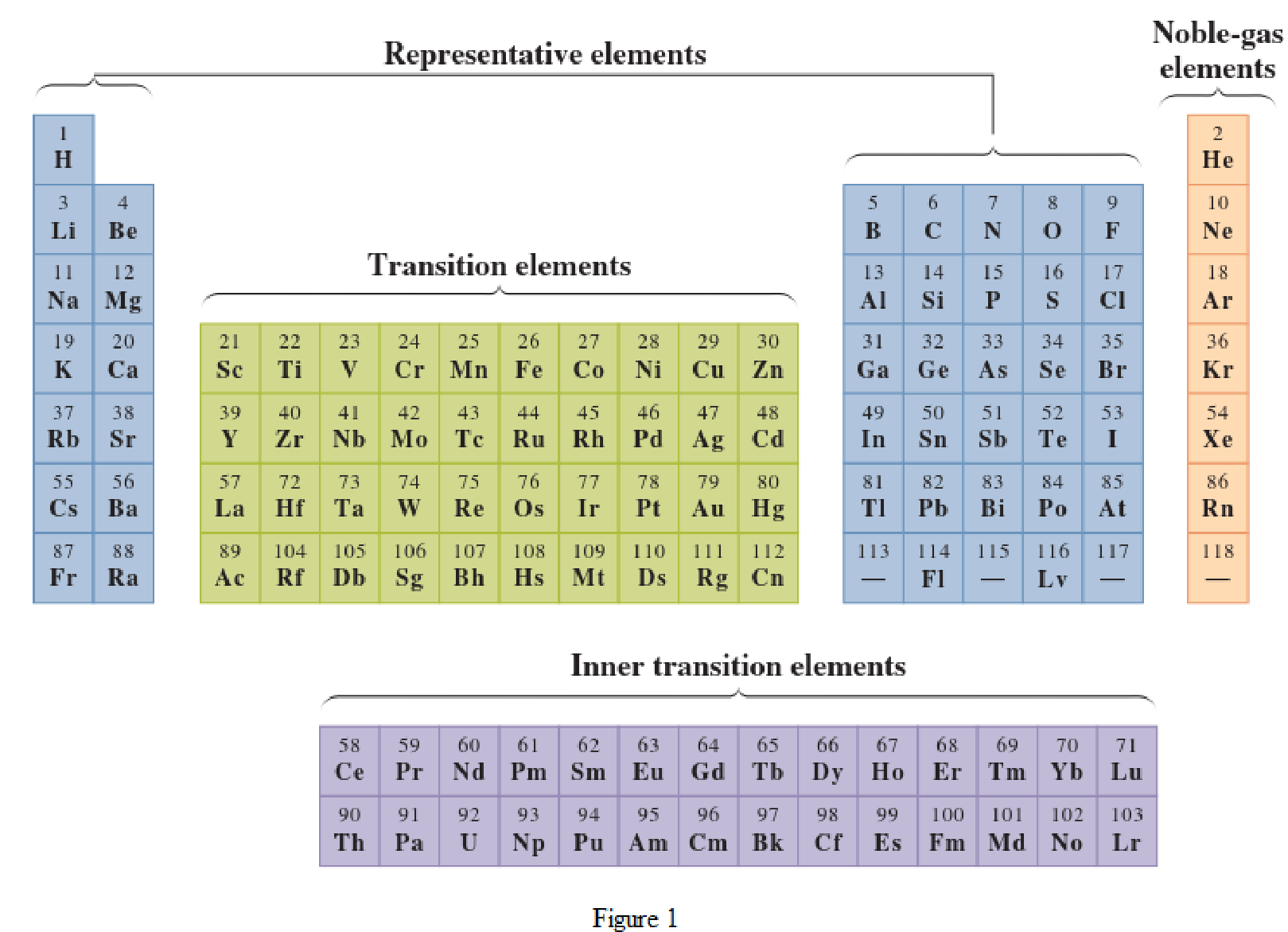
In the given periodic table, how many elements those are highlighted which represent nonmetallic representative elements have to be determined.
Concept Introduction:
Elements in the periodic table are classified in several different ways and out of them two most common systems are,
- System based on the physical properties in which they are classified as metals and nonmetals.
- System based on electronic configuration in which they are classified as noble-gas, representative elements, transition elements, or inner-transition elements.
Noble-gas elements are the ones that are located in far right of periodic table. The physical state of these elements at room temperature is gas. The noble gases have their electronic configuration ending with
Representative elements are the ones that are in s area and area of the periodic table. They have partially filled s subshell or p subshell in their electronic configurations. Some of the elements are nonmetals while others are metals.
Transition elements are the ones that are located in d area of periodic table. They have the distinguishing electrons in their d subshell. All the transition elements are metals.
Inner transition elements are the ones that are located in f area of the periodic table. They have the distinguishing electrons in their f subshell. All inner transition elements are metals.
(d)
Interpretation:
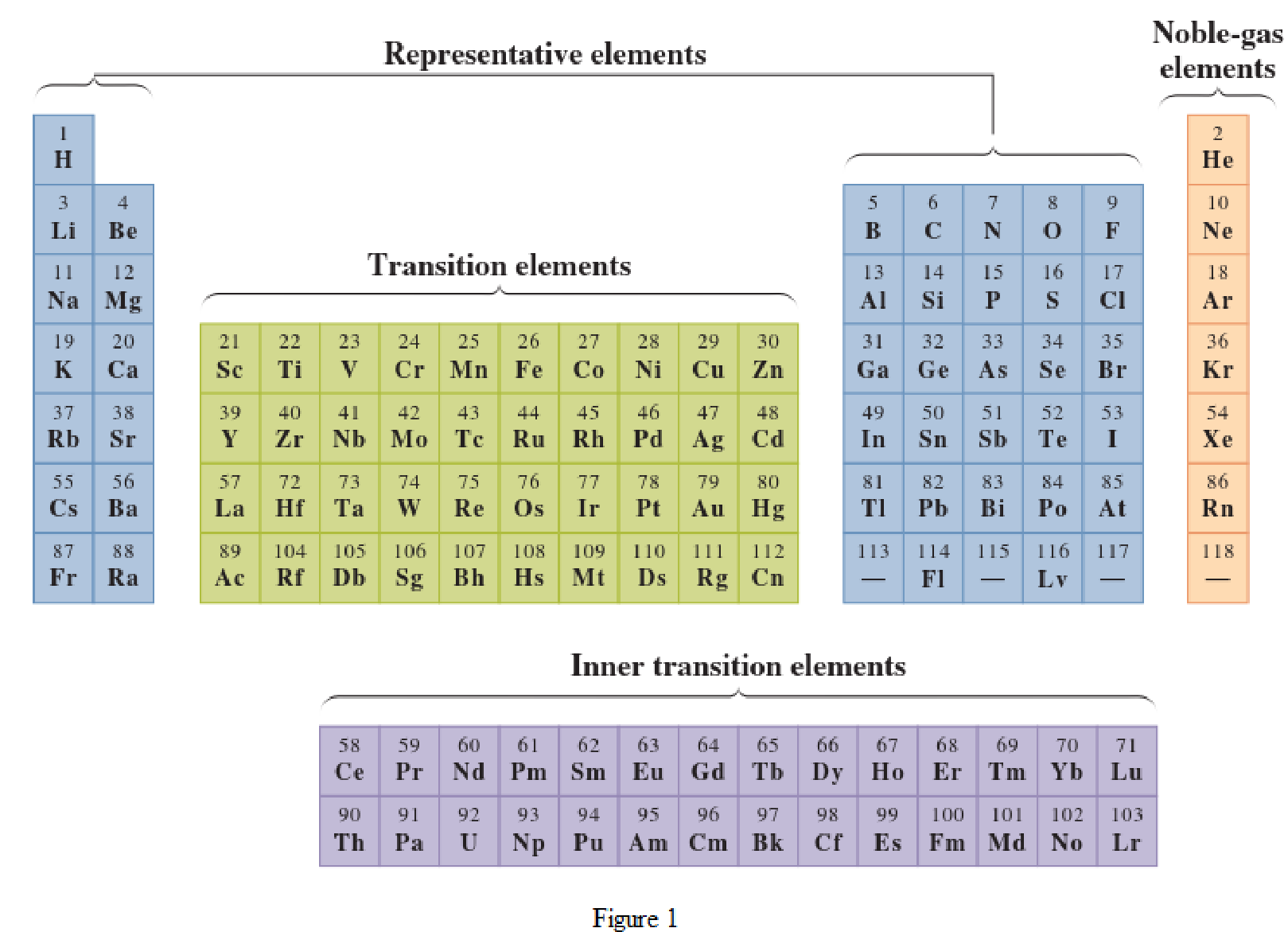
In the given periodic table, how many elements those are highlighted which represent metals have to be determined.
Concept Introduction:
Elements in the periodic table are classified in several different ways and out of them two most common systems are,
- System based on the physical properties in which they are classified as metals and nonmetals.
- System based on electronic configuration in which they are classified as noble-gas, representative elements, transition elements, or inner-transition elements.
Noble-gas elements are the ones that are located in far right of periodic table. The physical state of these elements at room temperature is gas. The noble gases have their electronic configuration ending with
Representative elements are the ones that are in s area and area of the periodic table. They have partially filled s subshell or p subshell in their electronic configurations. Some of the elements are nonmetals while others are metals.
Transition elements are the ones that are located in d area of periodic table. They have the distinguishing electrons in their d subshell. All the transition elements are metals.
Inner transition elements are the ones that are located in f area of the periodic table. They have the distinguishing electrons in their f subshell. All inner transition elements are metals.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General, Organic, and Biological Chemistry
- Copper (Cu) has an atomic number of 29 and a mass number of 64. One copper atom, therefore, has how many protons? a.27 b.29 c.31 d.35arrow_forward. The elements with atomic numbers of 93 or greater are referred to as the _________ elements.arrow_forwardThe atomic mass of lithium on a periodic table is 6.94u. Lithium has two natural isotopes with atomic masses of 6.10512u and 7.01600u. Calculate the percentage distribution between the two isotopes. Pure lithium is composed of two isotopes.arrow_forward
- The existence of isotopes violates one of the original ideas of Dalton’s atomic theory. Which one?arrow_forwardLabel the subatomic particles shown in Figure 4.22.arrow_forwardThe chemical name for limestone, a compound of calcium, carbon, and oxygen, is calcium carbonate. When heated, limestone decomposes into solid calcium oxide and gaseous carbon dioxide. From the names of the products, tell where you might find the atoms of each element after the reaction. How does Daltons atomic theory explain this?arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





