
Concept explainers
a)
Interpretation:
The group should be determined to which Z belongs in the following Lewis structure and along with an example of such a compound or ion which actually exists.
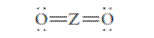
Concept Introduction:
Number of lone pair and bond pair electrons on an atom can give the number of valence electrons. This is indicated by the group number of an atom.
Group number is equal to the valence shell electrons of an atom or electrons present in the outermost shell. Valence shell electrons of the atom take part in the bonding.
Formal charge on an atom is defined as the charge assigned to the atom in a molecule. It is assigned to an atom or molecule by assuming that the electrons in
Formal charge is equal to
b)
Interpretation:
The group should be determined to which Z belongs in the following Lewis structure and along with an example of such a compound or ion which actually exists.
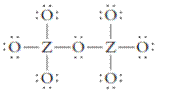
Concept Introduction:
Number of lone pair and bond pair electrons on an atom can give the number of valence electrons. This is indicated by the group number of an atom.
Group number is equal to the valence shell electrons of an atom or electrons present in the outermost shell. Valence shell electrons of the atom take part in the bonding.
Formal charge on an atom is defined as the charge assigned to the atom in a molecule. It is assigned to an atom or molecule by assuming that the electrons in chemical bonds are shared equally between atoms, apart from relative electronegativity.
Formal charge is equal to
c)
Interpretation:
The group should be determined to which Z belongs in the following Lewis structure and along with an example of such a compound or ion which actually exists.
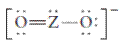
Concept Introduction:
Number of lone pair and bond pair electrons on an atom can give the number of valence electrons. This is indicated by the group number of an atom.
Group number is equal to the valence shell electrons of an atom or electrons present in the outermost shell. Valence shell electrons of the atom take part in the bonding.
Formal charge on an atom is defined as the charge assigned to the atom in a molecule. It is assigned to an atom or molecule by assuming that the electrons in chemical bonds are shared equally between atoms, apart from relative electronegativity.
Formal charge is equal to
d)
Interpretation:
The group should be determined to which Z belongs in the following Lewis structure and along with an example of such a compound or ion which actually exists.
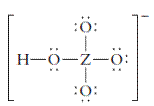
Concept Introduction:
Number of lone pair and bond pair electrons on an atom can give the number of valence electrons. This is indicated by the group number of an atom.
Group number is equal to the valence shell electrons of an atom or electrons present in the outermost shell. Valence shell electrons of the atom take part in the bonding.
Formal charge on an atom is defined as the charge assigned to the atom in a molecule. It is assigned to an atom or molecule by assuming that the electrons in chemical bonds are shared equally between atoms, apart from relative electronegativity.
Formal charge is equal to
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Principles of Modern Chemistry
- Following is a structural formula for guanidine, the compound by which migratory birds excrete excess metabolic nitrogen. The hydrochloride salt of this compound is a white crystalline powder, freely soluble in water and ethanol. Q.Write a Lewis structure for guanidine showing all valence electronsarrow_forwardWhich of these atoms cannotserve as a central atom in a Lewis structure: (a) O; (b) He; (c) F; (d) H; (e) P? Explain.arrow_forwardwrite the lewis structure of the molecule including the resonance forms, and the formal charges. the relative positions of the atoms are given as references answer all or i will downvotearrow_forward
- Draw Lewis diagrams for the following compounds. In the formula the symbol of the central atom is given first. (Hint: The valence octet may be expanded for the central atom.) (a) PF5 (b) SF4 (c) XeO2F2arrow_forwardComplete the rest of the table for N, O or X by drawing the number of Lewis structures specified.arrow_forwardOn the basis of the electronegativity values given in Fig. 12.3. indicate which is the more polar bond in each of the following pairs. msp;a.OClorOBrc.PSorPOb.NOorNFd.HOorHNarrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





