
EBK LIFE: THE SCIENCE OF BIOLOGY
11th Edition
ISBN: 8220103935432
Author: Sadava
Publisher: MAC HIGHER
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3.1, Problem 5R
Summary Introduction
To review:
The reasons for which the following structures of galactose and mannose are considered as isomers, and the type and properties of the
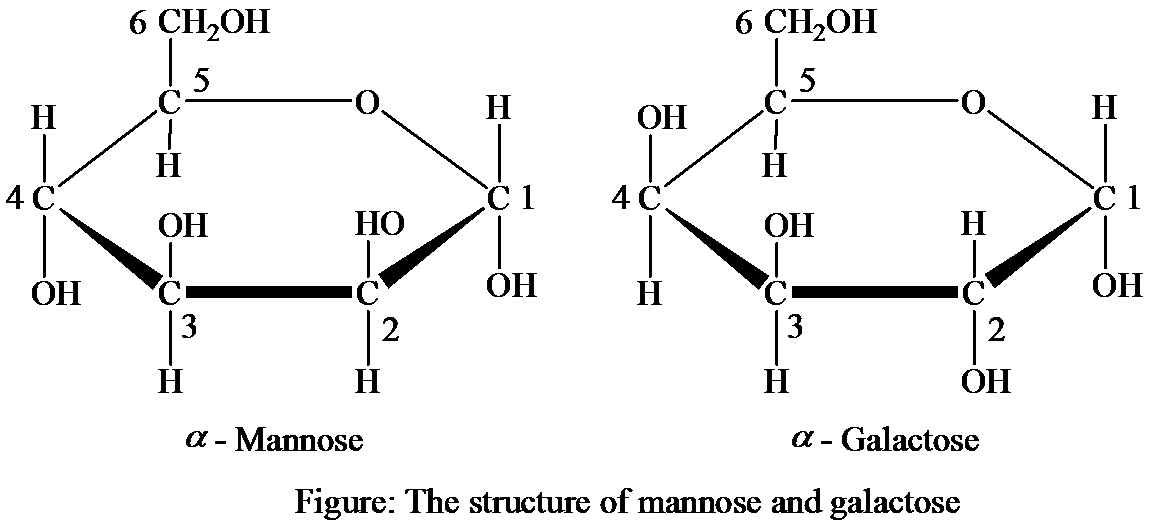
Introduction:
Isomers are those compounds, which have same chemical formula, but different orientations of atoms and molecules. Mannose and galactose are isomers since the chemical formula for both are same. Both of them form the structural isomers.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What are the main functional groups present in carbohydrates? Illustrate and explain.
How is the cyclic formula formed in glucose or fructose?
What is the position of the glycosidic bond in maltose, sucrose, lactose?
What is the importance of osazone formation?
What is invert sugar?
Classify polysaccharides.
Show linoleic and linolenic acid in numbers.
Compare the physical properties of saturated and unsaturated fats.
What are structural polysaccharides?
What are the lactose maltose sucrose glycoside bond drawings and galactose glucose mannose shapes?
Which of the following are a pair of structural isomers?
Glucose and Mannose
Galactose and Fructose
Ribulose and Xylulose
ribose and glucose
The correct answer is Galactose and Fructose. Please explain why and discuss how to identify structural isomers.
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Olestra has been used in certain snack foods as an alternativeto fats and oils. Its structure consists of a sucrose molecule inwhich all free hydroxyl groups have formed esters with oleicacid (an 18-carbon monounsaturated fatty acid). Olestra molecules contain no calories because they are exceptionallylarge and cannot be digested. Draw the structure of olestra.Use R—COOH as an abbreviation for oleic acid.arrow_forwardIndicate whether each of the following statements about the structural characteristics of monosaccharides is true or false. a. Both an aldehyde group and a ketone group are always present. b. An aldehyde group and at least two hydroxyl groups must be present.arrow_forwardDo structural polysaccharides typically have extensive hydrogen bonding?arrow_forward
- Compare/contrast the structures of the following carbohydrate pairs: (a) D-ribose and D-ribulose (b) isomaltose and maltose (c) cellulose and amylosearrow_forwardWhat are lipids and how can lipids be identified based on their physical propeties? How are lipids derived? What are the elements of Lipids?arrow_forwardIdentify the component monosaccharides of each of the following compounds and describe the type of glycosidic linkage in each.arrow_forward
- what is a monosaccharide? differentiate the aldoses and ketoses of monosaccharides and draw structures of glucose and fructose.arrow_forwardWhen blood levels of glucose are higher than normal,glucose molecules react with protein side chains such as thatof lysine residues to produced glycated side chains. Explainwhy this process is dangerous. What is the name of the firstfunctional group that is formed during glycation?arrow_forwardWhich of the following statements regarding lipids is most accurate? 1. Lipids are synthesized by ribosomes 2. Saturated fats tend to be solid at room temperature because of the polar hydrocarbon chains 3. Saturated fats tend to be liquid at room temperature due to hydrogen bonding 4. Polyunsaturated fats tend to be liquid at room temperature due to numerous double bonds in the hydrocarbon chains 5. The empirical formula for lipids is typically C1H201arrow_forward
- Differentiate the class of carbohydrates and their chemistry Compare and contrast proteoglycans, glycoproteins, and glycosaminoglycans. Cite examples for eacharrow_forwardWrite the reaction equation for the formation of sucrose, indicate the bonds that connect the monosaccharides in the disaccharide. Biological value of sucrose.arrow_forwardExamine the membrane lipid pictured below and answer the following questions: a. Is this lipid classified as a phospholipid or a glycolipid? Explain your answer in two sentences.b. Is this lipid considered a sphingolipid or a glycerophospholipid? Justify your answer in two sentences. c. What fatty acid chains are used in this lipid? Are they saturated or unsaturated? What functional group enables them to connect to the backbone?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license