
EBK LIFE: THE SCIENCE OF BIOLOGY
11th Edition
ISBN: 8220103935432
Author: Sadava
Publisher: MAC HIGHER
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3.3, Problem 2R
Summary Introduction
To review:
The presence of several hydrogen bonds and the strong structure in the given Figure 1, representing the linear structure of cellulose after referring to its molecular structure shown in Figure 2.
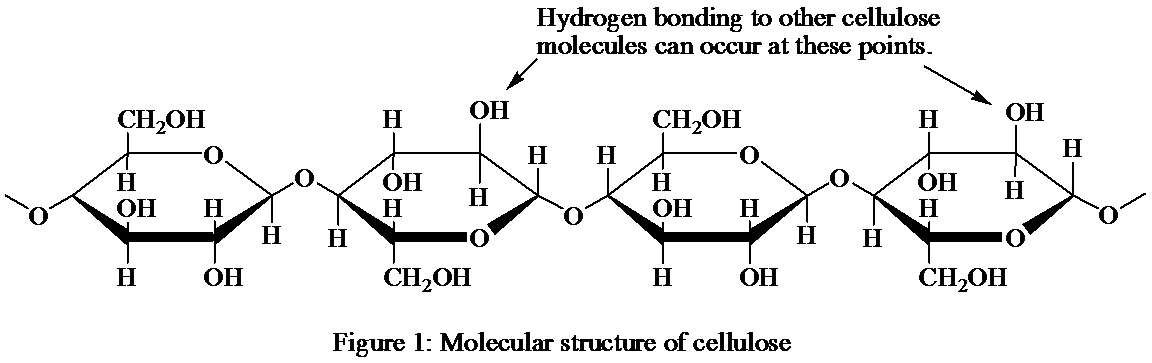
Figure 2: Molecular structure of cellulose
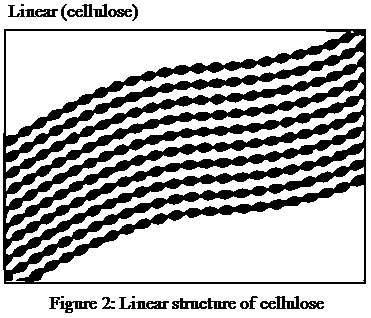
Figure 1: Linear structure of cellulose
Introduction:
Cellulose forms an important component of the plant cell walls. The cellulose protein is present in abundance on the earth. It is the polysaccharide, which is composed of the glucose. The glucose units found in the cellulose are majorly linked by the beta-glycosidic linkages unlike in glycogen and starch, where these units are found linked by alpha-glycosidic linkage.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The structure below shows that of a trisaccharide that is composed of (going from top left to bottom right) galactose, glucose, and fructose.
More specifically, the component monosaccharides are [D-galactose, L-galactose], [D-glucose, L-glucose], and [D-fructose, L-fructose] .
Help me
H
OH
CH2OH
D
H
ОН
H
الحزن
ОН
CH2OH
ОН
H
ОН I
Which of the following statements correctly describes this structure?
(A) The monomer units are bonded by beta 1-2 glycosidic linkage.
B
The monomer units are bonded by alpha 1-2 glycosidic linkage.
The monomer units are bonded by alpha 1-4 glycosidic linkage.
The monomer units are bonded by beta 1-4 glycosidic linkage.
H
CH2OH
H
ОН
H
ОН
H
ОН
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Consider the structure of the tripeptide (in its fully protonated form) below. H H H + I || H₂N-C-C-N-C-C-N-C-C-OH 1 I | H CH₂ H CH₂ CH₂ T C=O OH AA1 0=0 || HC-CH3 CH3 AA2 AA3 0=C 1. Give the sequence of the tripeptide using the ONE-LETTER DESIGNATION (UPPERCASE LETTER) with NO spaces and symbols between each letter. 2. How many ionizable groups are there in the tripeptide? Give the numerical value (e.g., 10 not ten). • pH 10: {Choices: -2, -1, 0, +1, +2} 3. Which amino acid residue has one ionizable group left upon forming the tripeptide? {Choices: AA1, AA2, AA3, none, all} 4. Give the net charge of the dominant structure of the tripeptide at the given pH values. The pK, values of the amino acids are given in Table 1. • pH 4: {Choices: -2, -1, 0, +1, +2}arrow_forwardHow many reducing ends and non-reducing ends will a oligosaccharide have with 4 exact branch points in its structure?arrow_forwardThe monosaccharide shown below polymerizes to form a polysaccharide where each sugar residue is linked to each other by an a (1– 6) linkage. Which of the choices below represents the polysaccharide just described? он он H H он но OH CH2OH CH2OH ÇH2OH H. OH OH OH H OH H OH H H H. OH Он OH H H OH H H. QH OH H. OH OH OH ÇH2OH ÇCH2OH CH2OH QH QH H. H OH H. OHarrow_forward
- Consider the structure shown below. он 3 5 CH; O CH, O H CH; O 1 H-N-CH,-C-N-CH;-C-N-CH-C-N-CH-C-N-CH-C-ơ 2 H. H H Fill in the blank with an integer (1, 2, 3, 4, 5..) as shown in the diagram or to represent a specified number. A hydrophilic side chain is indicated by the numberarrow_forwardWhat is the structure labeled 5 in the following image and explain why it’s correct.arrow_forwardThe structure shown is an example of a type of macromolecule (carbohydrate, lipid, protein, or nucleic acid) that is an important biological polymer. Identify the type of macromolecule macromolecule in the cell. H H H NH H H H H H N|H shown, the type of monomer it is made from, how you came to your conclusion and the general types of important biological functions of this type of 2=0 N-H -O-Harrow_forward
- In the structure in the picture, the monosaccharide units (alpha-D-galactopyranosyl and beta-D-allopyranose) are linked via alpha-1->3 glycosidic bond. Then, the formed disaccharide units are linked via beta-1->4 glycosidic bond. Then, an Oligosaccharide is formed which has 10 monosaccharide units, meaning that it contains 5 disaccharide units. Question: Is this oligosaccharide a good substrate for glycolysis? Provide two reasons for your answer.arrow_forwardWhy do think nucleotides are also sometimes referred to as “bases” or “nucleotide bases”? What is the difference between a nucleotide and a nucleoside? Explain by giving an example, using structures. What kind of bond links a base to some sugar? Is the sugar an alpha or beta anomer? In what position(s) of the sugar does this occur? In what position(s) of the base does this occur? Instead of the term “Formation of a nucleoside”, what could the name of the reaction be? What functional group is being formed? Define the primary structure of DNA/RNA. Compare and contrast to the primary structure of proteins.arrow_forwardWhat type of glycosidic bond is shown below, α or β?arrow_forward
- Beta-carotene is red-orange pigment abundant in plants and fruits. Assuming that it can be modeled as a particle in a 1-D box, estimate how long the red-conjugated chain is given that A = 475 nm in hexane.' Using an average conjugated CC bond length of 140 pm, how long should the "box" be? Figure 1. Beta carotene %3Darrow_forwardGenAlex Medical, a little-known division of a major Swiss pharmaceutical firm, recently developed a new synthetic steroid S. Their patent application describes S as "made from a steroid nucleus with hydroxyl groups at positions 3 and 7, and methyl groups at positions 4 and 12." In the drawing space below, draw the chemical structure of S. If more than one structure is possible, you can draw any of them. Click and drag to start drawing a structure. ח' G c Earrow_forwardAn amylose chain is 5000 glucose units long. At how many places must it be cleaved to reduce the average length to 2500 units? To 1000 units? To 200 units? What percentage of the glycosidic links are hydrolyzed in each case? (Even partial hydrolysis can drastically alter the physical properties of polysaccharides and thus affect their structural role in organisms.)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license