
Concept explainers
(a)
Show the cubic direction
(a)
Explanation of Solution
Show the cubic direction
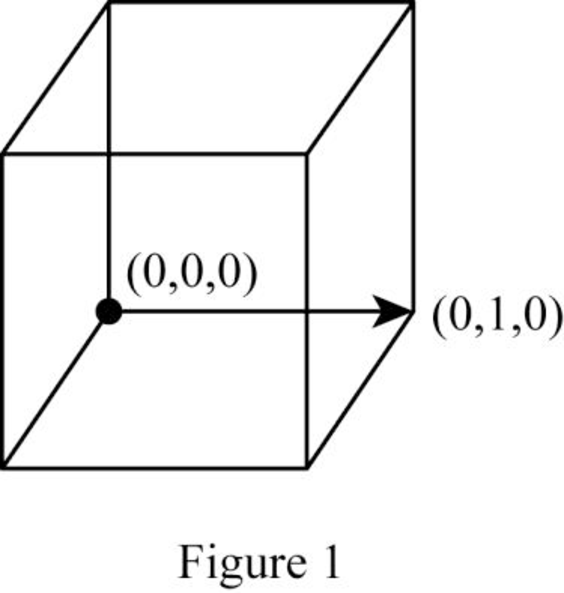
Position coordinates:
For the cubic direction
Repeat distance:
For the cubic direction
(b)
Show the cubic direction
(b)
Explanation of Solution
Show the cubic direction
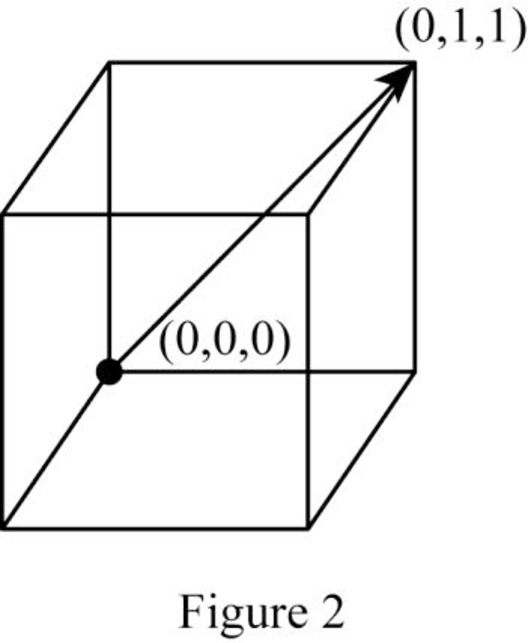
Position coordinates:
For the cubic direction
Repeat distance:
For the cubic direction
(c)
Show the cubic direction
(c)
Explanation of Solution
Show the cubic direction
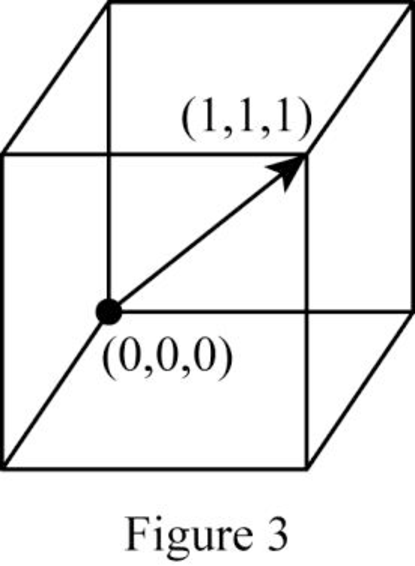
Position coordinates:
For the cubic direction
Repeat distance:
For the cubic direction
(d)
The angle between the cubic directions
(d)
Answer to Problem 29AAP
The angle between the cubic directions
Explanation of Solution
Write the expression to calculate angle between the cubic directions
Here, Miller indices of the cubic plane 1 are
Conclusion:
Substitute 0 for
Thus, the angle between the cubic directions
Want to see more full solutions like this?
Chapter 3 Solutions
Foundations of Materials Science and Engineering
- Determine the Miller indices of the cubic crystal plane that intersects the following position coordinates(points): (1, ½, 1), (½, 0, ¾), and (1, 0, ½). Sketch the plane, and show all steps and distances.arrow_forwardIf the atomic radius for Pb- 0.175nm and the crystal structure is FCC, calculate the atoms numbers in the unit cell, find the relation between atomic radius and lattice constant and what is the atomic packing factorarrow_forwardShow how to get this indices miller bravais for a HCP structure, right from the 3 indices used on cubic structures (hkl).arrow_forward
- In a cubic crystal, draw any three (3) parallel planes and determine their miller indices.arrow_forwardWhat are all the planes in {101} family of an orthorhombic lattice?arrow_forwardDetermine the Miller indices for the planes of the cubic unit cell shown in figure 2, according to the coordinate axis system shown.arrow_forward
- Determine the possible crystal structure of Zinc Blende structure (ZnS), if the radii are as follows Zn+2 = 0.074 and S-2 = 0.184.arrow_forwardDetermine the Miller Indices of Direction C and D in an HCP crystal.arrow_forwardGallium has an orthorhombic structure,with a0 5 0.45258 nm, b0 5 0.45186 nm,and c0 5 0.76570 nm. The atomic radiusis 0.1218 nm. The density is 5.904 g/cm3,and the atomic weight is 69.72 g/mol.Determine(a) the number of atoms in each unit cell;and(b) the packing factor in the unit cell.arrow_forward
- Molybdenum has an atomic radius of 0.145nm. The volume of its cubic unit cell is0.0375 nm3 . What is the geometry of the molybdenum unit cell?arrow_forwardWhat are the three types of lattice structures?arrow_forwardWith a hexagonal crystal structure where a0=0.22858nm, co=0.35842nm, an atomic radius of 0.1143nm, density of 1.848g/cm3, and an atomic weight of 9.01g/mol, what is a)number of atoms in each unit cell and b) the packing factor in each unit cell.arrow_forward
 Understanding Motor ControlsMechanical EngineeringISBN:9781337798686Author:Stephen L. HermanPublisher:Delmar Cengage Learning
Understanding Motor ControlsMechanical EngineeringISBN:9781337798686Author:Stephen L. HermanPublisher:Delmar Cengage Learning
