
Concept explainers
(a)
Interpretation:
The Lewis structure for an aldetetrose is to be drawn.
Concept introduction:
Monosaccharides are called simple sugars, with the number of oxygen atoms same as the number of carbon atoms. The general formula for monosaccharides is
Answer to Problem 4.25P
The Lewis structure for an aldetetrose is
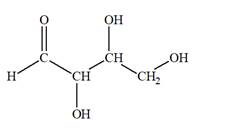
Explanation of Solution
Aldotetrose is a mono-saccharide. The name “aldo” indicates that the carbonyl group must be present at the terminal carbon atom, while the word “tetrose” indicates a chain of four carbon atoms. Thus, aldotetrose is a sugar having an
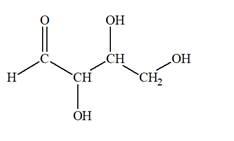
The molecular formula for the above structure is
Altetrose is a mono-saccharide having a carbonyl group at the terminal carbon atom, and its molecular formula is
(b)
Interpretation:
The Lewis structure for a ketotetrose is to be drawn.
Concept introduction:
Monosaccharides are called simple sugars, with the number of oxygen atoms same as the number of carbon atoms. The general formula for a monosaccharide is
Answer to Problem 4.25P
The Lewis structure for a ketotetrose is
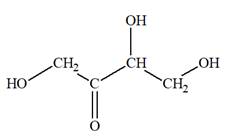
Explanation of Solution
Ketotetrose is a type of mono-saccharide. The name “keto” indicates that the carbonyl group must be present at the internal carbon atom, while the word “tetrose” indicates the chain of four carbon atoms. Thus, ketotetrose is a sugar having a carbonyl group at the internal carbon atom, and the structure must have four carbon atoms. The general molecular formula for mono-saccharides is
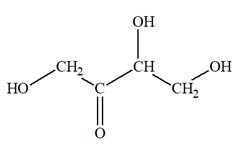
The molecular formula for the above structure is
Ketotetrose is a mono-saccharide having a carbonyl group at the internal carbon atom, and its molecular formula is
(c)
Interpretation:
The Lewis structure for an aldetriose is to be drawn.
Concept introduction:
Monosaccharides are called simple sugars, with the number of oxygen atoms same as the number of carbon atoms. Thus, the general molecular formula for a monosaccharide is
Answer to Problem 4.25P
The Lewis structure for an aldetriose is
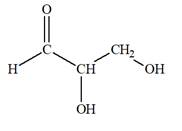
Explanation of Solution
Aldotriose is a type of mono-saccharide. The name “aldo”’ indicates that the carbonyl group must be present at the terminal carbon atom, while the word “triose” indicates a chain of three carbon atoms. Thus, aldotriose is a sugar having an aldehyde group at the terminal carbon atom, and the structure must have three carbon chain. The general molecular formula for mono-saccharides is

The molecular formula for the above structure is
Altetrose is a mono-saccharide having a carbonyl group at the terminal carbon atom, and its molecular formula is
(d)
Interpretation:
The Lewis structure for a ketotriose is to be drawn.
Concept introduction:
Monosaccharides are called simple sugars, with the number of oxygen atoms same as the number of carbon atoms. Thus, the general molecular formula for a monosaccharide is
Answer to Problem 4.25P
The Lewis structure for a ketotriose is
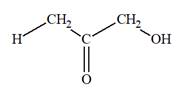
Explanation of Solution
Ketotriose is a type of mono-saccharide. The name “keto” indicates that the carbonyl group must be present at the internal carbon atom, while the word “tetrose” indicates the chain of four carbon atoms. Thus, ketotriose is a sugar having a carbonyl group at the internal carbon atom, and the structure must have three carbon atoms. The general molecular formula for mono-saccharides is

The molecular formula for the above structure is
Ketotetrose is a mono-saccharide having a carbonyl group at the internal carbon atom, and its molecular formula is
(e)
Interpretation:
The Lewis structure for a ketohexose, which is different from fructose, is to be drawn.
Concept introduction:
Monosaccharides are called simple sugars, with the number of oxygen atoms same as the number of carbon atoms. Thus, the general molecular formula for a monosaccharide is
Answer to Problem 4.25P
The Lewis structure for a ketohexose, which is different from fructose, is
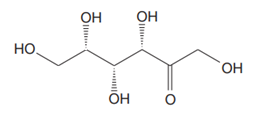
Explanation of Solution
The Lewis structure for fructose is
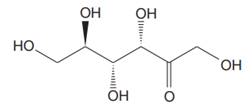
Fructose is a ketohexose with a molecular formula

The molecular formula for the above structure is
Ketohexose is a mono-saccharide having a carbonyl group at the internal carbon atom, and its molecular formula is
Want to see more full solutions like this?
Chapter 4 Solutions
Organic Chemistry: Principles And Mechanisms: Study Guide/solutions Manual (second)
- Draw the R group of (a) glycine; (b) isoleucine; (c) tyrosine.arrow_forwardcan you explain how both those carbons are chiral in the second molecule, like what are the four different groups that those carbons are attached to?arrow_forwardA) Define "reducing sugar." (b) Sucrose is a disaccharide composed of glucose and fructose (Glc(1 → 2)Fru). Explain why sucrose is not a reducing sugar, even though both glucose and fructose are.arrow_forward
- Just little explanation Asap Thanksarrow_forwardArabinoxylan is a copolymer of arabinose and xylose-two five-carbon sugars. A portion of the polysaccharide is shown here. (a) Identify and classify each glycosidic linkage. (b) Based on the structure, do you think arabinoxylan functions as a storage polysaccharide or a structural polysaccharide? Explain your reasoning. HOH,C. OH Но OH OH но но OH Но LO. Но HOH,C HOH2C НО OH OH CH,OHarrow_forwardTextbook Problem Which of the terms glycerophospholipid, sphingophospholipid, and sphingoglycolipid applies to each of the following characterizations? More than one term may apply, or none of the terms may apply in a given situation a. Membrane lipid b. Saponifiable lipid c. Amide linkage present d. Has a "head and two tails" structurearrow_forward
- How many constitutional isomers of acyclic monosaccharides are there that can be classified as aldohexoses? As ketohexoses?arrow_forwardWhich statement about saccharides is correct? (A). Alpha anomers are more common than beta anoomers. (B). Two monosaccharides can form a disaccharide and oxygen. (C). Polysaccharides with alpha or beta glycosidic linkages have the different properties. (D). Disaccharides can be cyclic or acyclic.arrow_forwardProblem: (a) Draw six (6) constitutional isomers of C6H1202. (b) Which of the compounds you drew would be the most soluble in water? Explain your choice. (c) Which of the compounds you drew would be the most soluble in hexane? Explain your choice.arrow_forward
- Draw structures for the following molecules. (a) 3-bromo-2-nitropentane; (b) 2,2-dichloro-4,4,5-trinitroheptane;(c) 1,2,3,4-tetranitrobutane; (d) 6-iodo-1,2-difluorohexanearrow_forwarda) Draw structural formulas for the two monosaccharides that result when structure III istreated as shown below. (b) Draw the monosaccharide that results when product A from the previous step (Q1a) istreated with Br2/H2O. (c) Draw the monosaccharide that results when product A from the previous step (Q1a) istreated with 1.NaBH4 / 2. H2O.arrow_forwardExample: Consider the naturally occurring amino acid, alanine, and its enantiomer.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning  Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div





